Vietnam
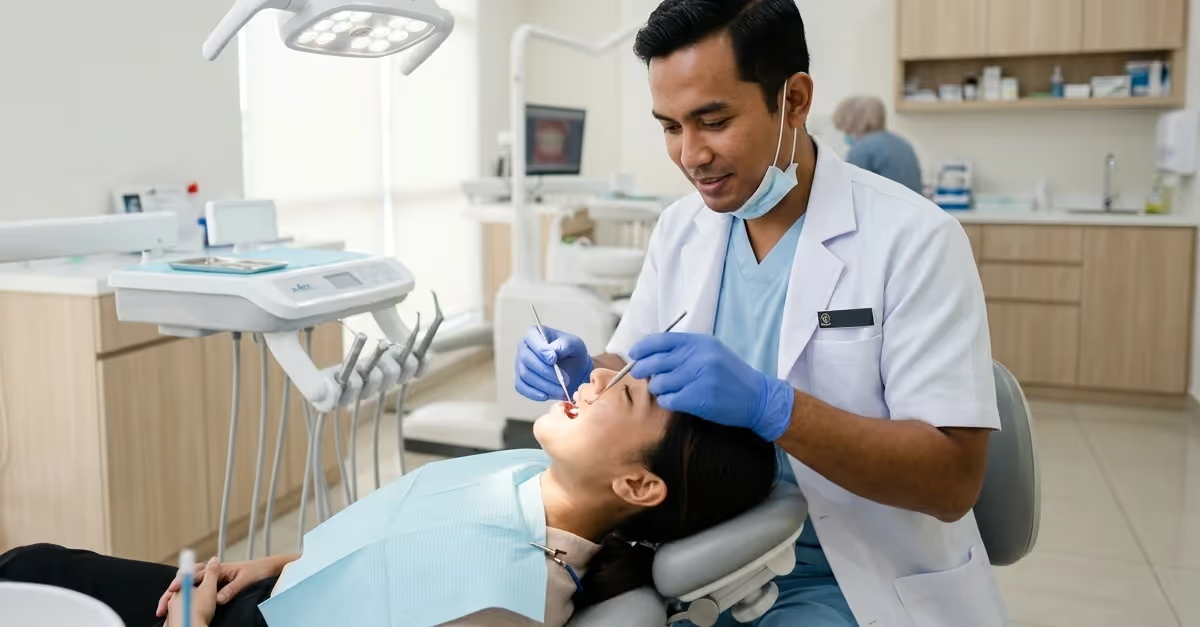
Enter Southeast Asia’s dental market with a clear strategy: navigate regulatory complexity, sequence market entry, and retain control of registrations to scale efficiently.
.avif)
Learn about Cisema’s redesigned website with faster performance, clearer navigation and expanded Asia Pacific coverage.

From registration to tariffs and CPTPP benefits, here’s what Japanese cosmetics manufacturers need to know about Vietnam’s cosmetics market.

Learn how to prepare a compliant dossier for Vietnam medical device registration in this guide for foreign manufacturers.

Learn how to manage medical device renewals, variations, and change notifications in Vietnam with step-by-step MOH submission guidance.

Learn about license validity and renewal rules for medical devices in Southeast Asia.

Explore Vietnam’s fast-track and standard medical device registration pathways and the role of reference approvals in shortening timelines.

Explore fast-track medical device registration in Southeast Asia, cut timelines by 6–12 months, and accelerate market entry with Cisema.

Cosmetic product registration and notification in Vietnam involves specific processes, fees, and timelines under the ASEAN Cosmetic Directive.

Learn how to fast-track medical device registration in ASEAN: timelines, costs, and license options in Malaysia, Singapore, and Vietnam.

Gain an overview of the medical device registration process in Vietnam with timelines, fees, and Authorized Representative requirements.

Vietnam Assigns MOH Oversight for Chemicals in Medical Products
Vietnam's Decree No. 26/2026/ND-CP” confirms MoH oversight of chemicals in medical devices, IVDs, and medicines.

Vietnam Strengthens Enforcement of Cosmetic Notification and PIF Compliance
Vietnam’s DAV intensifies inspections through March 2026, targeting cosmetic notification compliance and Product Information File readiness.

Vietnam Transfers High-Risk Medical Device Evaluation to Specialized Authorities
Vietnam’s Ministry of Health has delegated medical device oversight to specialized agencies, impacting registration and post-market surveillance.

Vietnam Cuts Advertising Red Tape for Healthcare Products
Vietnam’s MoH recently published Circular drops several healthcare advertising regulations and streamlines requirements for many categories.

Vietnam Draft Cosmetic Decree Signals Tougher Compliance Environment
Vietnam’s draft cosmetic management decree would replace existing rules and strengthen post-market controls for cosmetic companies.

Vietnam Authorities Step Up Cosmetic Recalls as Enforcement Tightens
Vietnam’s Drug Administration (DAV) is stepping up cosmetic product recalls as post-market surveillance tightens.

Vietnam Consolidates Medical Device Rules Under Decree 98 Framework
Vietnam’s Ministry of Health issued “Consolidated Document No. 08/VBHN-BYT (2026)”, unifying medical device regulations.

Vietnam Delays Implementation of New Health Supplement Regulations
Vietnam temporarily suspends implementation of stricter registration and import inspection rules for health supplement foods.

Vietnam Formalizes Electronic Medical Device Procedures Under Decision 3830/QĐ-BYT
Vietnam’s Ministry of Health moves to standardize and digitize medical device administration, improving transparency and compliance.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
