Your Partner for Regulatory Affairs, Quality Compliance, Clinical Research & Certification Support Across China & APAC
Cisema delivers expert clinical development, regulatory, and market access services to help companies bring their products to market faster and stay compliant across Asia Pacific. Whether pharmaceuticals, medical devices, cosmetics, health foods, toys, machinery, or electronic components, we support your success from development to approval and beyond.
With deep experience in China and a growing presence across the region, we provide local insight with global perspective, enabling innovation, ensuring regulatory compliance, and accelerating your growth in target markets.



Our Story
Cisema was founded in 2002 by Stefan Fischer, who first worked in Beijing with Siemens in the 1990s. When China joined the World Trade Organisation (WTO) in 2001, Stefan recognized a clear need: companies wanting to access the Chinese market needed a trusted partner who understood both regulatory requirements and cross-cultural dynamics.
Cisema was created to meet that need.
Today, we are a trusted provider of market access services for companies entering China and the wider Asia Pacific region. From startups to multinational leaders, we guide clients through regulatory strategy, product registration, independent testing, and post-market compliance with precision and care.
Our offices span China’s industrial hubs and strategic locations in the APAC region. We combine on-the-ground knowledge with international quality standards, delivering bilingual support that bridges regulatory gaps and drives faster time to market.
We remain proudly family-owned and focused on building long-term relationships. With leadership experience in China dating back to 1985, we offer unmatched depth of regulatory intelligence, cultural fluency, and expertise.
Whether launching a new product, expanding your regional footprint, or ensuring compliance, Cisema’s market access services provide the clarity and confidence you need to succeed in global markets.
Meet Some of Our Team

Hamish King

Stefan Fischer

Sunny Cao

Nick Guo

Tan Shouzheng

Chelsea Wang

Florian Waegele

Qiang Zhou
Our Approach
At Cisema, our approach is comprehensive and data-driven, designed to deliver actionable insights and measurable value for our clients.
We combine real-world evidence and regulatory expertise to develop tailored strategies that support every stage of market entry and product lifecycle management. Our team works closely with clients to assess their unique needs, ensuring that each solution is aligned with business objectives and regulatory requirements.
Local Focus
While global market access is essential for business expansion, achieving success in each country requires a deep understanding of local regulatory requirements, market conditions, and patient or client needs. Cisema’s team leverages local expertise and strong in-country partnerships to develop tailored strategies that address the unique challenges and opportunities of each market.
We work closely with clients to ensure that their products meet all local regulatory standards, supporting seamless market entry and ongoing compliance. By combining our global perspective with local knowledge, we help clients navigate regulatory complexities, deliver value to customers and consumers and drive sustainable growth in every market they enter.
Why Work with Cisema?
One Partner, Full Lifecycle Support
From pre-market strategy to post-market compliance, we provide support to internal regulatory teams and ensure seamless lifecycle management.
Regional Reach, Global Presence
Based in Asia and Europe with a global team delivering global reach.
Cross-Industry Expertise
Pharma, biotech, medical devices, cosmetics, supplements, veterinary, industrial and consumer goods.
Proven Results
Over 20 years of regulatory submissions, regulatory expertise, and approvals to drive business success.

Our Locations
Head Office
Cisema (Hong Kong) Limited
7/A Shun Pont Commercial Building
5-11 Thomson Road, Wan Chai
Hong Kong
Other Locations
Asia-Pacific: Beijing | Hangzhou | Tianjin | Qingdao | Singapore | Penang | Tokyo | Seoul | Bangkok | Ho Chi Minh City | Manila | Jakarta | Taipei
Europe: Munich
North America: Chicago
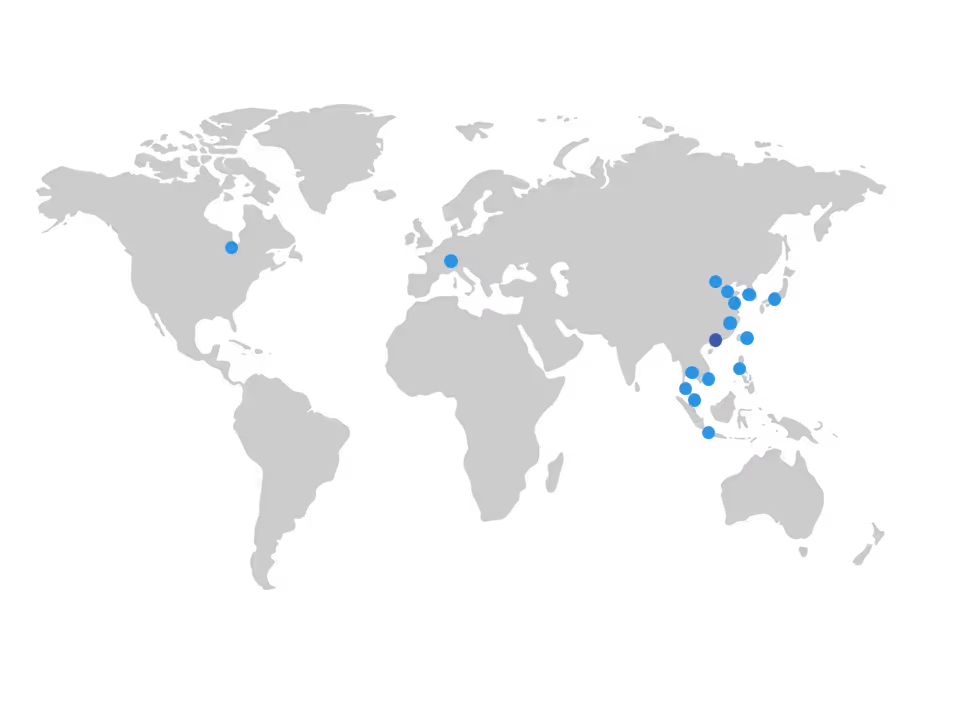
Our Global Network
Cisema connects global companies with China and Asia Pacific through a network of international offices. Our headquarters in Hong Kong serves as the central hub for our APAC operations and a primary point of contact for companies entering or expanding across the region. Our additional offices across the region strengthen our presence in key markets and provide direct access to local regulatory authorities, test laboratories, and industry partners.
Our multilingual teams in offices worldwide work across time zones to ensure efficient communication and responsive service. Through clear coordination, transparent timelines, and careful budget management, we aim to process client requests within 24/48 hours and keep projects progressing smoothly.
In China, our offices in Hangzhou, Beijing, Tianjin and Qingdao are strategically located to support efficient regulatory and testing processes. The Beijing office leads certification and regulatory coordination, while the Hangzhou office focuses on sourcing and supply chain support in collaboration with other branches. The Tianjin office is located near leading NMPA-certified test centers, enabling efficient coordination of local testing and compliance activities.
Together, this network connects Cisema directly to China’s major economic regions, including the Yangtze River Delta, the Pearl River Delta, and the Bohai Bay area, allowing us to maintain close communication with authorities, respond quickly to regulatory developments, and provide flexible operational support for our clients.
We also have increasing presence throughout other markets, including Malaysia, Singapore, Vietnam, Indonesia, the Philippines, Thailand, Taiwan and South Korea. These enable us to serve our clients throughout East Asia and the ASEAN economies and act as local authorised representative so that manufacturers don't need to set up their own offices or rely on distributors, who are focused on sales targets rather than obtaining approvals, complying with regulations (to keep the sales won) and guarding intellectual property.
Our long-standing relationships with regulatory authorities and accredited laboratories enhance efficiency and help anticipate regulatory developments. This proactive approach enables us to keep clients and industry partners informed and prepared for changes across China and the wider Asia Pacific region.
Across all offices, secure digital infrastructure and modern IT systems ensure that project data remains protected while allowing seamless collaboration between our teams and clients worldwide.






