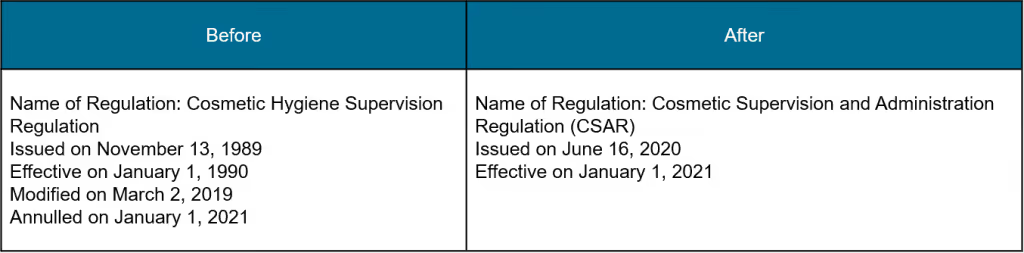
Cosmetics

The NMPA announced various updates regarding soap, toothpaste, hair removal, hair growth, breast beauty, body slimming and deodorizing products.
The NIFDC released 7 technical draft proposals for detecting chemical agents and the evaluation of efficacy claims in cosmetic products in China.

Since the implementation of the CSAR (Cosmetics Supervision and Administration) was officially announced in June 2020, China has been preparing for the regulation to become effective on January 1, 2021. In November 2020, the NMPA (National Medical Products Administration) released 5 regulatory updates to support the CSAR. We have summarised these below.
Draft instructions for cosmetic registration and filing dossiers
In order to implement the CSAR, this draft includes a total of 6 chapters and 57 articles to further clarify and refine the specific information requirements for cosmetics registration and filing.
The CSAR requires the manufacturers to register their special cosmetic products with hair coloring, hair perming, freckle whitening, sunscreen, anti-hair loss functions or with new efficacy claims, whilst filing would be required for general cosmetics (in the past referred to as non-special cosmetics).
The public consultation period is open until November 30, 2020.
Draft instructions for new cosmetic ingredient registration and filing
This draft includes a total of 28 articles addressing the requirements for the registration and filing for new cosmetic ingredients:
- Registrants are responsible for the safety monitoring of the new cosmetic ingredients and required to submit an annual report for three years
- Registration will be required for new cosmetic ingredients with higher risk functions such as antiseptics, sun protection, hair coloring, freckle removal, whitening, etc., while filing will be required for other cosmetic ingredients
The public consultation period is open until November 30, 2020.
Draft guidelines for cosmetic efficacy claim evaluation
This draft consists a total of 22 articles to provide technical guidance on the evaluation of efficacy claims for the cosmetics industry. Different types of efficacy claims require their own evaluation method and are identified in the draft to ensure their scientific character and accuracy.
The public consultation period is open until November 30, 2020.
Draft measures for the management of cosmetic supplementary testing methods
This draft consists of 6 chapters and 29 articles covering the scope, procedures, guidelines and rules of cosmetic supplementary testing methods. Such supplementary testing methods will be applied for sampling inspections, quality and safety investigations and adverse reaction investigation.
The public consultation period is open until December 10, 2020.
Draft administrative measures on toothpaste products
This draft consists of 23 articles addressing the requirements of ingredients, product filing and efficacy evaluation of toothpaste products:
- If the toothpaste product uses a new ingredient for the first time in the market, the registrant must ensure that a mandatory technical standard is implemented before marketing. If there is a standard already, only the safety evaluation report of the ingredient would be required for submission
- The NMPA will formulate the classification catalogue for efficacy claims and keep it updated in a timely manner
- There should be sufficient and reliable scientific evidence to support the efficacy claims
- The product labelling should not claim to have medical effects in an expressive or implicit manner
- Any violations including, but not limited to, the following will be punished:
- Not registering or filing the product as required
- Illegal use of other ingredients
- Violation of labelling requirements
- Not verifying the efficacy claims as required
The public consultation period is open until December 12, 2020.
If you are interested in making a public submission, please share your opinions with us before the consultation period ends.
By Jacky Li. Contact Cisema if you would like to learn more.

4 draft regulations were released to support the implementation of the CSAR whilst the safety and technical standards for cosmetics will be amended.

China has carried out a series of measures to support the CSAR (Cosmetic Supervision and Administration Regulation) and the cosmetics regulatory framework.

On July 31, 2020, the Beijing MPA (Medical Products Administration) made an announcement to implement stricter supervision on medical cosmetology products.

On June 29, 2020, China’s State Council announced the implementation of the new regulation for CSAR (Cosmetics Supervision and Administration), which will become effective on January 1, 2021.
Major changes
Background
The definition of cosmetics
Classification of Cosmetics
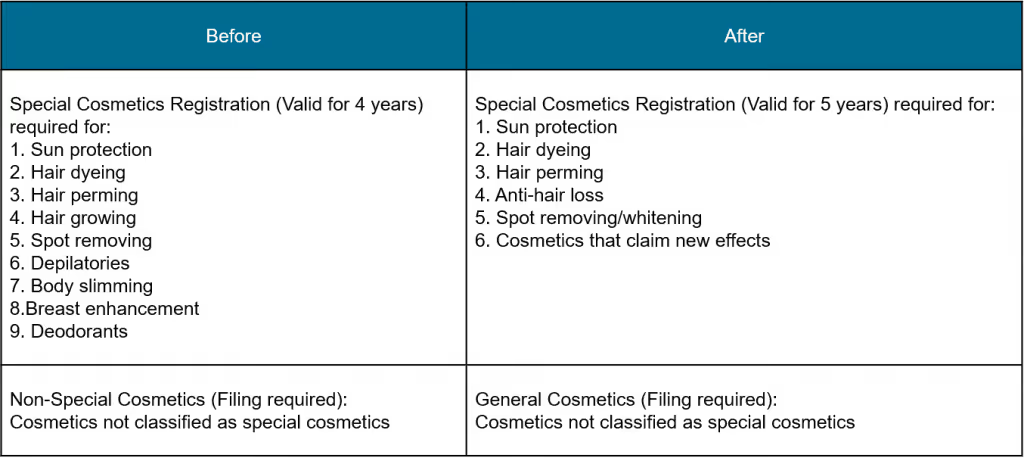
Note: Registered products with functions for hair growing, depilatories, breast enhancement, body slimming, and deodorants will no longer be classified as special cosmetics after a 5-year transition period.
Classification of Cosmetic Ingredients

Note: After the registration / filing of new ingredients, the registrant / filer should report the current status of consumer use and safety to the NMPA annually for 3 consecutive years. If any safety issues should occur during these three years, the registration / filing will be cancelled, otherwise, the new ingredient will be included in the list of used ingredients (IECIC, also known as Inventory of Existing Cosmetic Ingredients in China).
Toothpaste management

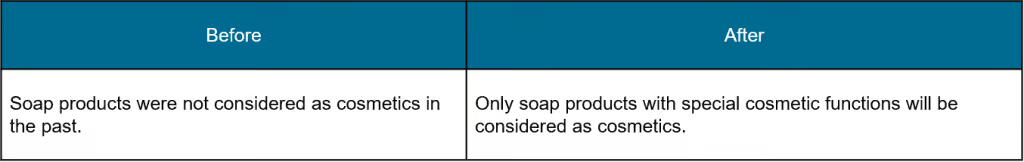
Note: According to the China Oral Care Industry Association’s announcement on July 2, 2020, toothpaste products are not considered as cosmetics. Further information will be disclosed by the NMPA.
Soap management
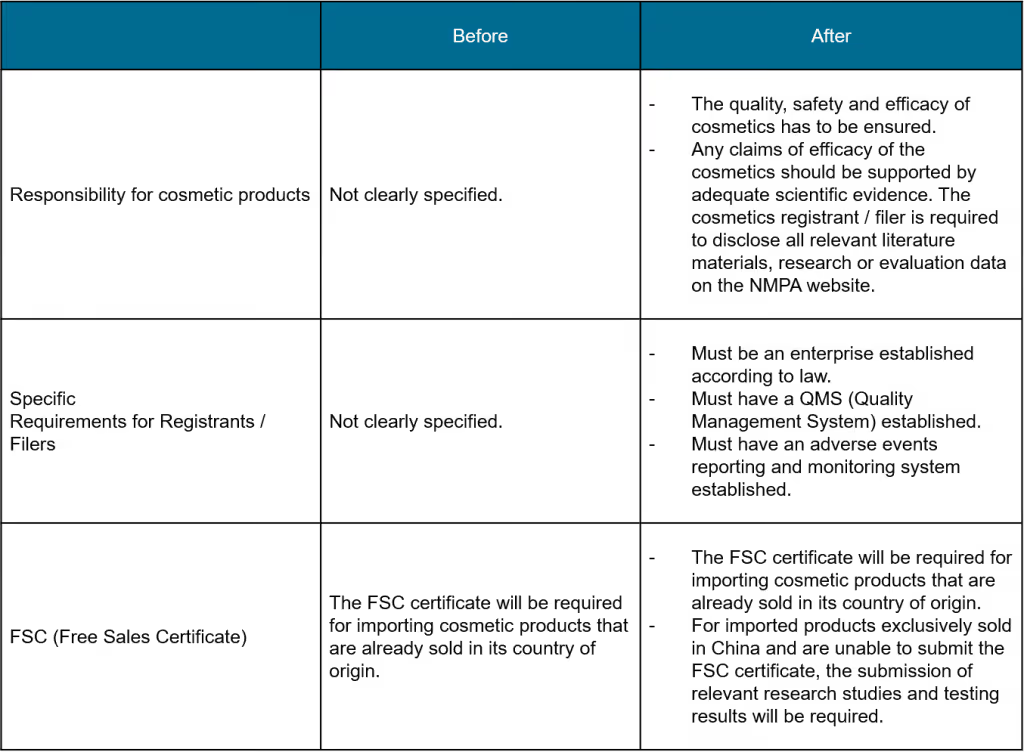
Requirements for registrants and filers
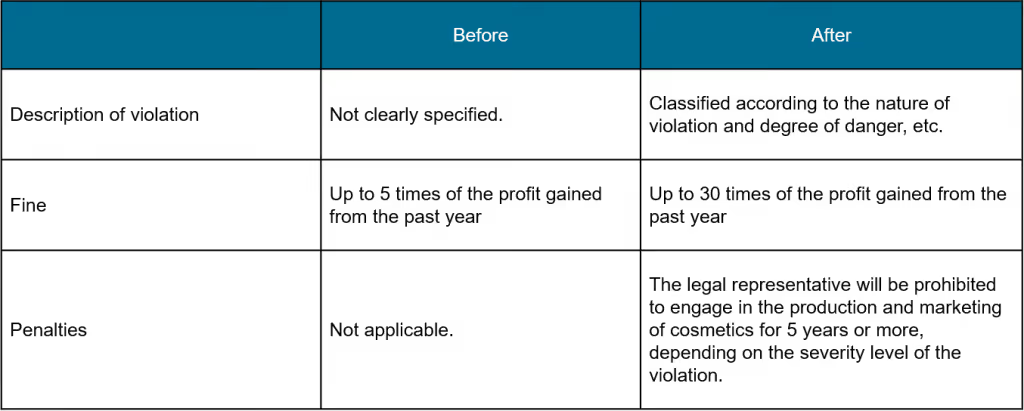
Penalties for violations
By Jacky Li. Contact Cisema if you would like to learn more.

Advantage Austria together with Ms. Anna King from CISEMA inform about the recent regulatory and institutional developments in China.

The Israel Export Institute co-hosted a webinar together with Cisema to introduce the China administration bodies, regulations and standards for cosmetics.

On 06.11.2019, the China NDRC (National Development and Reform Commission) announced (No.20-2019) the prohibition of daily chemical articles containing plastic microbeads for production after December 31, 2020, and for sale after December 31, 2022.Microbeads are small plastic particles, mostly included in cosmetic and cleaning products, and are known to cause water pollution to the marine and freshwater environments. Currently, 14 states have already taken action towards the ban on microbeads. Although the details of the ban for production and sales in China is still subject to further refinement, brands and manufacturers are expected to reformulate their products containing microbeads, while some have already replaced microbeads with natural abrasives as an alternative in daily chemical articles.The announcement (No.20-2019) is based on the Industrial Structure Adjustment Guidance Catalogue (2019 Edition) that was approved at the 2nd committee meeting on 27.08.2019, and has replaced the 2011 version since 01.01.2020.

On 22.11.2019, China's NMPA (National Medical Products Administration) announced a draft regulation concerning overseas inspection for cosmetic products, and launched the public consultation to receive feedback.According to the proposed regulation, it aims to introduce, standardize and optimize the inspection process. Once adopted, inspections on foreign manufacturers are required to be conducted by Chinese authorities from NMPA, which includes on-site inspections for any overseas facilities involved with the manufacturing process of cosmetic products that are intended to be sold in the China market.China NMPA will continue to solicit public opinions until 22.12.2019.

On 10.09.2019, the NMPA (National Medical Products Administration) announced changes to the procedures regarding cosmetics sample tests in China (No. 72-2019), followed by an interpretation of their announcement on 12.09.2019. Laboratories - that perform sample tests for cosmetics NMPA approval - need to re-apply for their qualification as a testing institutions because the relevant qualification will be automatically suspended on 01.11.2019. Until then - during the transitional period - there are two possible methods for managing tests. Cosmetics enterprises can follow the original procedures offline, as well as apply for sample tests online. Starting on 01.11.2019, however, laboratories have to be newly accredited. Furthermore, cosmetics enterprises and their NMPA Legal Agents will only be able to apply for and manage tests, which are related to cosmetics registration/filing, online.1) The goal is to improve the testing workflow, to standardize the test project requirements, to improve the efficiency of tests, and to provide strong technical support for cosmetics safety supervision.2) The prerequisite for testing institutions is to submit relevant information and obtain a new certification from the CMA (China Inspection Body and Laboratory Mandatory Approval).3) New guidelines for testing institutions include that testing must not be subcontracted, quality management system has to be ensured, testing work procedures for cosmetics registration and documentations have to be standardized, the facilities and equipment have to meet requirements.4) In order to improve work efficiency, an “Inspection Information System” was introduced to optimize the workflow of testing institutions. Via the online system, cosmetics manufacturers select a testing institution, submit relevant product information and apply for testing. The testing institution can then accept the test application and issues the test report online after testing.5) Changes in managing test samples include:Firstly, rather than regulatory authorities performing the on-site test sample sealing, the testing insitution seals the test samples. This change only applies to domestic “special” cosmetics.Secondly, laboratories accept samples produced for the purpose of testing if the product is not sold yet. (Please note, however, that the NMPA requires a product to be already sold in the country of origin.)6) The test requirements are changed to emphasize the safety of products during evaluation and some test items will be adjusted to better reflect the safety situation of products; for instance, by measuring dioxane.7) During the transition period – until 31.10.2019:Firstly, testing institutions should apply for their qualification because on 1 November, the old ones will be terminated.Secondly, already approved cosmetics, whose tests do not comply with the new specifications anymore, have to undergo supplementary testing a year of the announcement.Third, cosmetics manufacturers can follow the old (offline) way of applying for and managing tests, as well as the new “online” way.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
