Medical Devices

NMPA conducted China medical device supervision inspections on 11 different categories and announced the sampling results.

CMDE has been exploring the use of Real-World Evidence to support clinical evaluation since 2018 and is of increasing importance.
China’s NMPA has released draft guidelines for writing medical device IFU and seek for Industry stakeholders' comment until December 1, 2023.

Breakfast seminar on health technology in Hong Kong and GBA will be carried out on November 27, 2023, at the Finland Chamber of Commerce.
Medical devices urgently needed for clinical use in China have new draft requirements announced by the CMDE for temporary importation.

China medical device sampling inspection results have recently been announced by the NMPA on September 28, 2023

China medical device master filing aims to protect confidential information from being disclosed during the NMPA registration process

China launches an Expert Consultation Mechanism to solve interpretation issues of the updated GB9706 Medical Electrical Equipment Standards.
Human factors design for medical devices in China has draft guidelines revised and issued by the CMDE on October 10, 2023.
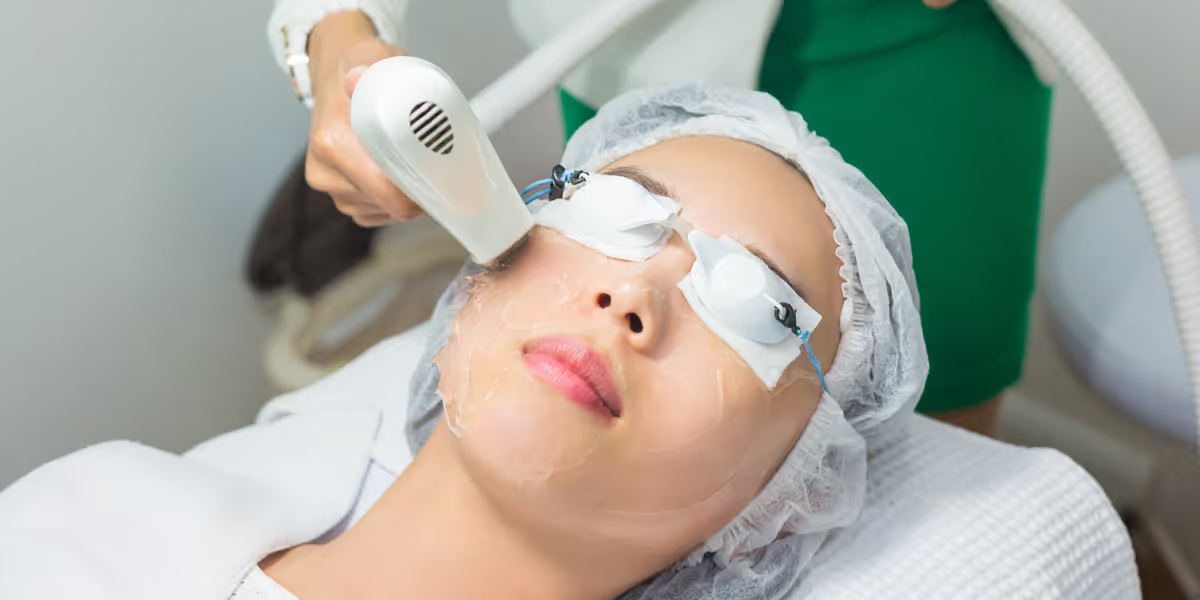
Clinical evaluation of laser therapy devices has new guidelines issued by the China Center for Medical Device Evaluation in July 2023.
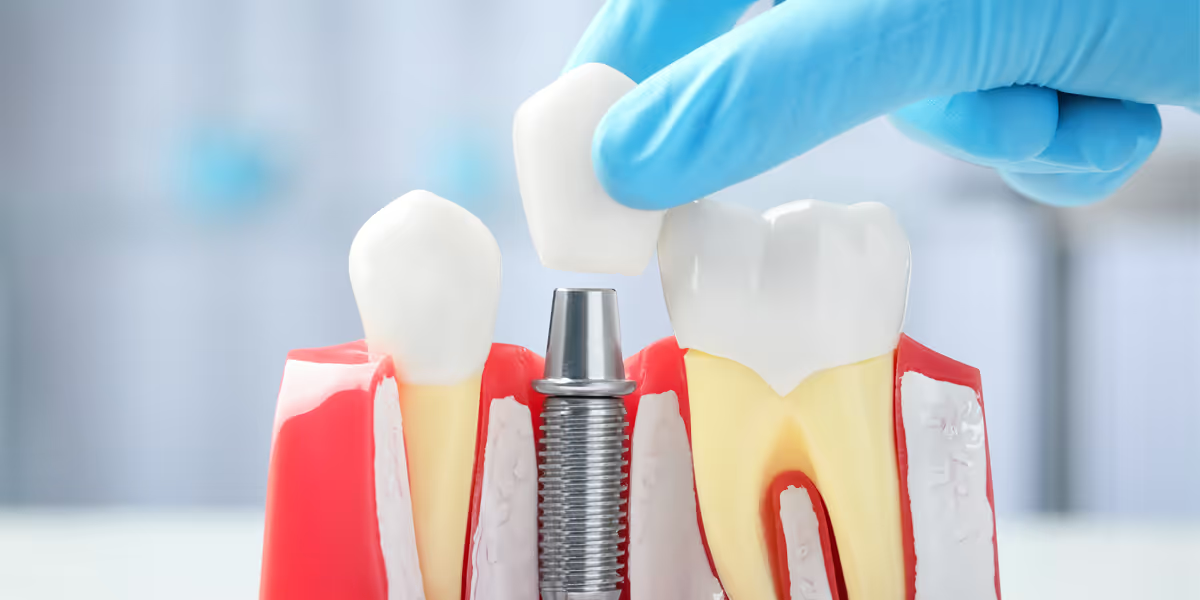
Clinical evaluation of dental implant systems has new guidelines issued by the China CMDE on August 14, 2023.

China Medical Device Webinar will be featuring speakers from U.S. Embassy Beijing, U.S. Patent and Trademark Office (PTO), IQVIA, and Cisema
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
