Medical Devices

Registration of Blood Dialysis Concentrates in China shall refer to the technical review guidelines revised by the CMDE on April 28, 2023.
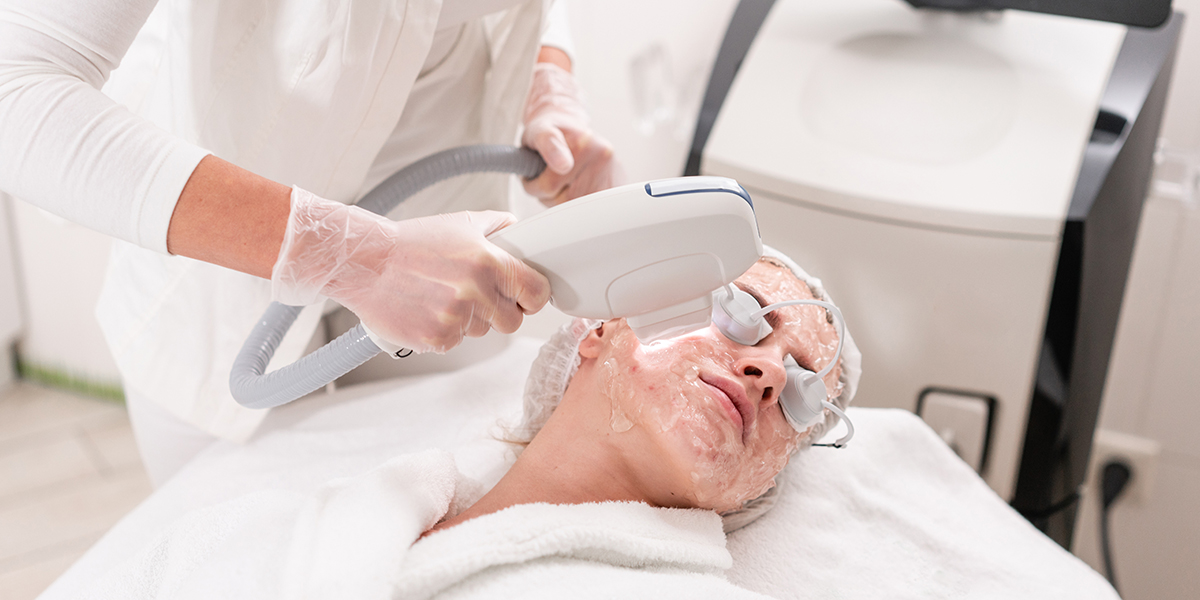
Intense pulsed light therapy device registration in China shall refer to the newly revised technical review guidelines issued in April 2023

Clinical trials of sodium hyaluronate complex solution have a set of draft guidelines issued by the China CMDE on May 17, 2023.
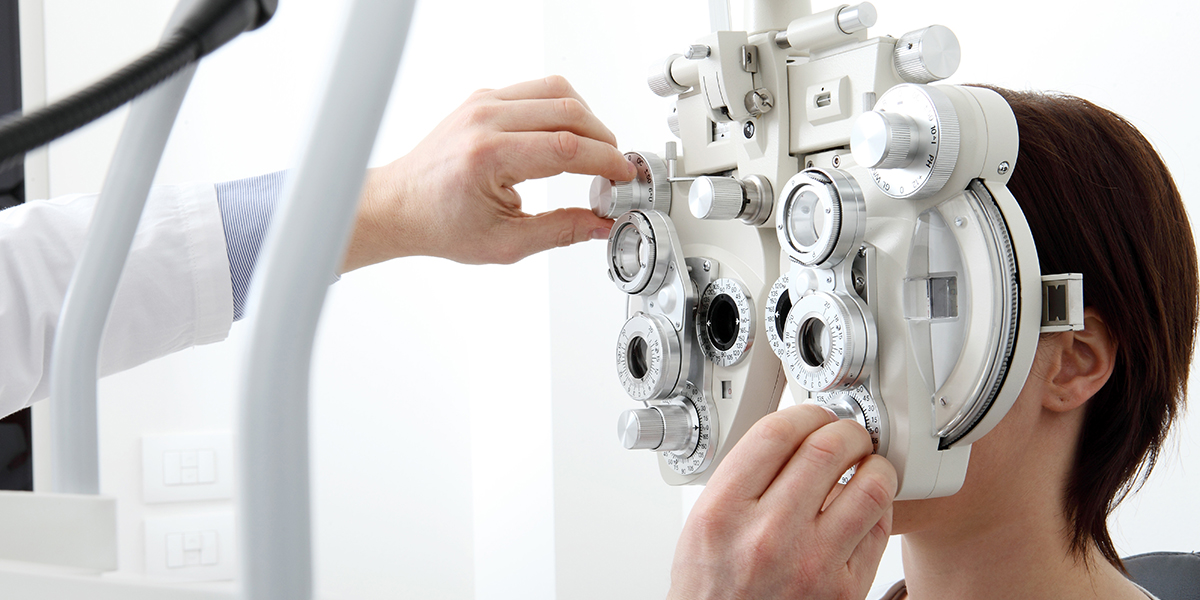
China registration of ophthalmic optical measurement devices shall refer to the respective technical review guidelines issued by the CMDE

Pre-communication for Hainan medical device clinical RWD applications will be required to follow the CMDE trial measures.
Guidelines for medical device registration in China have new development plans released in April 2023 - 108 will be implemented and revised

China medical device market eRPS data error correction matters will be incorporated into the business scope on April 20, 2023.

China medical device sampling inspection plan for 2023 covers 68 products including electronic endoscopes, medical oxygen concentrators etc.
China radio frequency beauty devices have new technical review guidelines issued to ensure that products are safe and effective
China’s NMPA issues further explanations on how to interpret and implement GB9706.1-2020 standards for medical electrical equipment

China releases a whitepaper on its AI medical device market. The AI medical device industry is becoming a new growth driver.

Ultrasound soft tissue surgical equipment in China have new technical review guidelines amended for registration dossier preparation
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
