Medical Devices
Medical device classification catalogue changes to non-sterile liquid/paste dressings, neurological/cardiovascular surgical instruments
Supervision of medical device distribution in China will be strengthened when the new regulatory measures take effect on May 1, 2022.

Supervision of medical device manufacturing in China will be strengthened when the new regulatory measures comes into effect on May 1, 2022.

The China Clinical Trials GCP (Good Clinical Practice) 2022 amended version will come into effect on May 1, 2022.
Importing drugs and medical devices from Hong Kong have been approved and amounted up to 15 products into the GBA as of March 2022.

2022 sampling inspection plan covers 60 products mainly targeting IVD reagents, anti-epidemic medical supplies, etc.

The NMPA report on China medical device standards management provided a comprehensive overview of the past year’s developments.

China issued digital health regulatory guidelines for registration of medical device software including AI, and for cyber security.

Medical device product technical requirements have new guideline issued by the China NMPA on February 9, 2022.
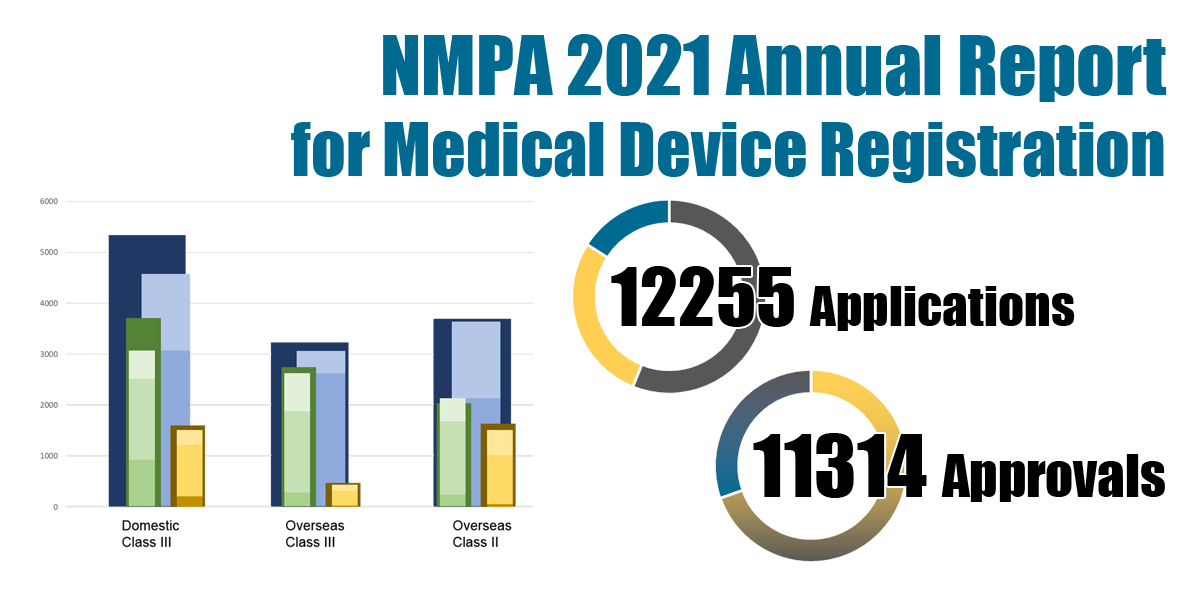
The NMPA reported a total number of 12,255 applications and 11,314 approvals for medical device and IVD registrations in 2021.
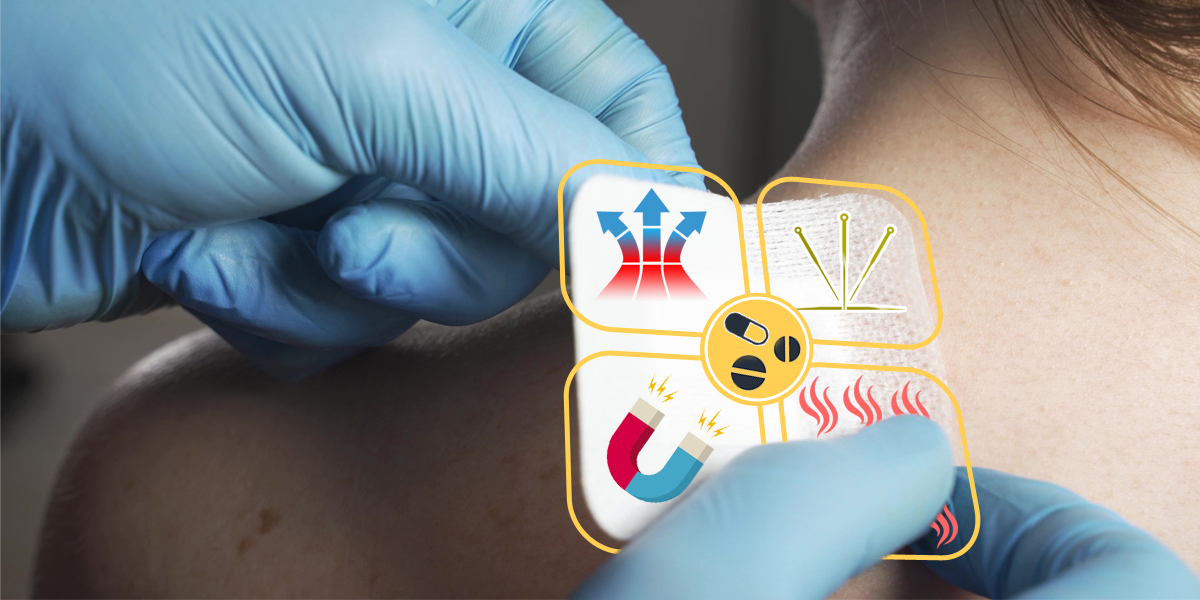
In order to restrain the illegal use of drug ingredients in medical dressing patches, the China NMPA approved the testing methods propose....

China Medical Device Standards have been recorded with a total of 1,849 as of December 2021, whilst 181 standards were newly issued in 2021.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
