Medical Devices

China innovative medical devices special review results have been announced by the NMPA on October 18, 2024.

China NMPA has announced a suspension of import, sale and use of laser surgery equipment manufactured by Jeisys Medical Inc.

Micro-volume syringe priority review application has been announced by the Center for Medical Device Evaluation on October 18, 2024.

Absorbable hemostatic product and other 11 medical devices registration review guidelines have been revised and published by China's CMDE.

The results of China’s latest round of medical device inspection were published by China’s NMPA on 11 September 2024.

InvestHK recently invited Cisema to share regulatory insights at their seminar event, highlighting opportunities in Hong Kong and GBA.

China’s CMDE has published medical device real-world data terms and definitions in draft format for industry comments by October 8, 2024.

China’s NMPA has released a draft Medical Device Administration Law to replace the current China Medical Device Regulations.
China’s NMPA has issued the requirements for the Temporary Import of Class II and Class III Medical Device for Urgent Clinical Need.

New Greater Bay Area regulations for importing Hong Kong drugs and medical devices starting December 1, 2024 will reduce review time
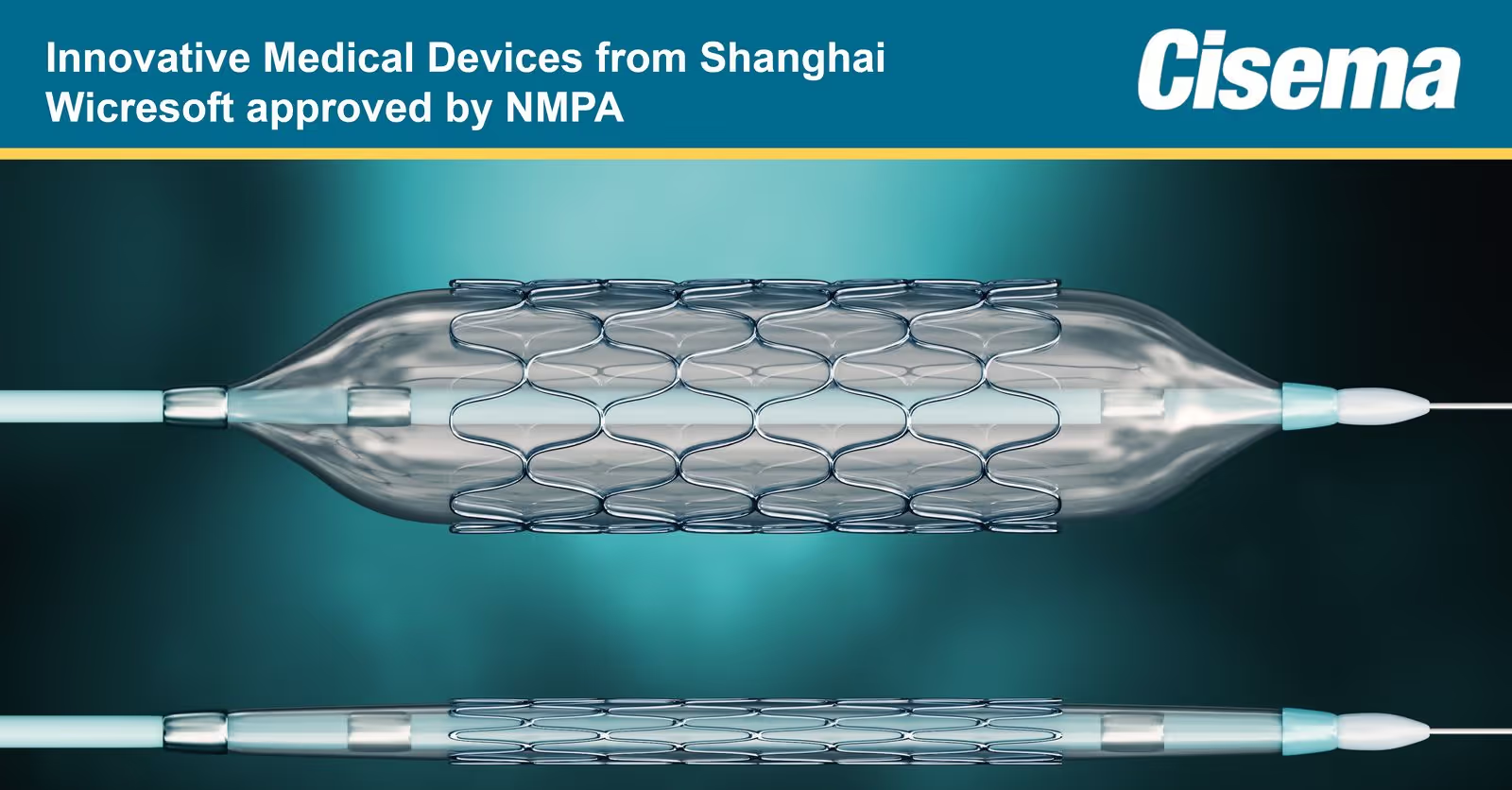
China’s NMPA has approved Shanghai Wicresoft’s Bioabsorbable Rapamycin Eluting Coronary Stent System through the Innovative Medical Device Green Pathway Programme.
China’s NMPA has announced a suspension of import, sale and use of two medical devices manufactured by SAMO S.p.A. because they failed to comply with regulatory requirements.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
