Pharma & Biotech

China NMPA regulations supporting Good Manufacturing Practice (GMP) of drugs in clinical trials will be implemented on July 1, 2022
The 14th Five-Year Plan for Drug Supervision Network Security and Information Construction issued by the NMPA will focus on 16 missions

Measures for the Supervision and Administration of Drug Production was issued by the SAMR and came into effect on July 1, 2020.

Measures for the Administration of Drug Registration took effect on July 1, 2020, to reform the drug review and approval system.

The Guideline for Pharmacovigilance Inspection urge MAHs to implement the main responsibility of pharmacovigilance.

China Drug Annual Reports Administrative Regulations was issued by the National Medical Products Administration on April 12, 2022.
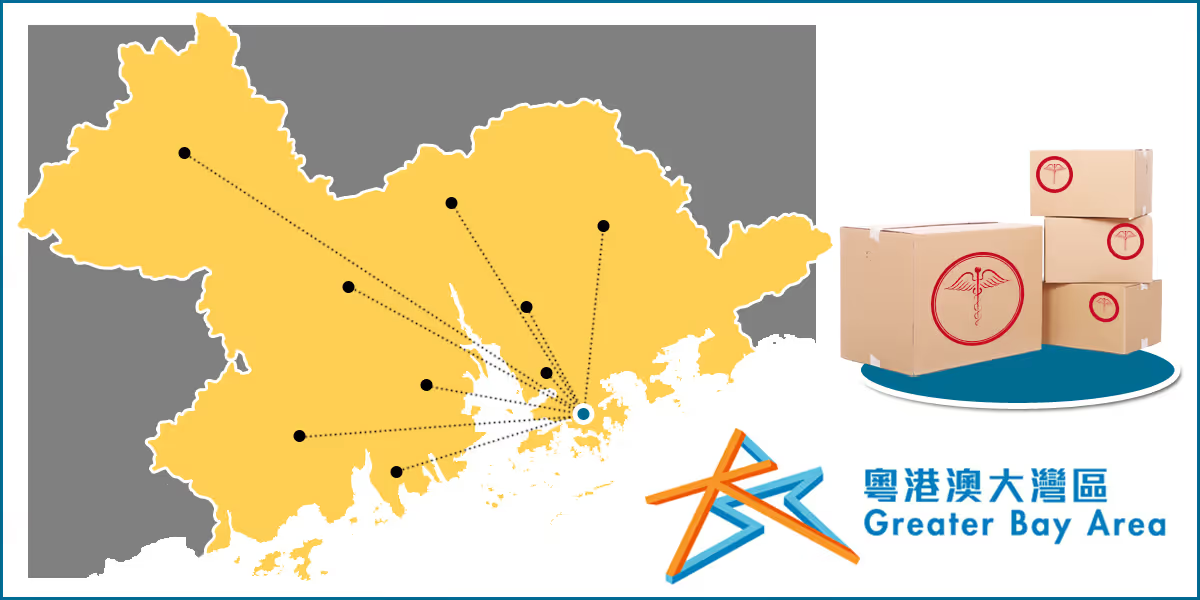
Importing drugs and medical devices from Hong Kong have been approved and amounted up to 15 products into the GBA as of March 2022.

Clinical trials GMP management specification first issued its draft in July 2018. The appendix draft was issued on January 18, 2022.

Acceptance and examination of chemical APIs trial guidelines have had its first draft issued in April 2020.

The NMPA published the 14th Five-Year Plan for National Drug Safety and Promotion of High Quality Development setting goals to (i) achieve...

The CDE published the trial working procedures for initiating drug verification and testing with immediate effect from Jan 1, 2022, onwards.

A variety of consultation channels are offered by the CDE to enhance the support of technical guidance and services to applicants.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
