Indonesia

Understand Indonesia halal certification requirements for health supplements, including BPJPH registration, ingredient review, and halal labeling obligations.

Learn how Indonesia halal cosmetic certification works, including BPJPH requirements, halal labeling rules, ingredient compliance, and registration obligations.

Learn which products are covered by Indonesia's halal framework, the labeling and manufacturing obligations, and phased compliance timelines.

Learn how to register medical devices in Indonesia, including Authorized Representative rules, classification, dossiers, timelines, and fees.

Cisema offers regulatory insight and market access strategy for medical devices in Thailand, Indonesia, and the Philippines.
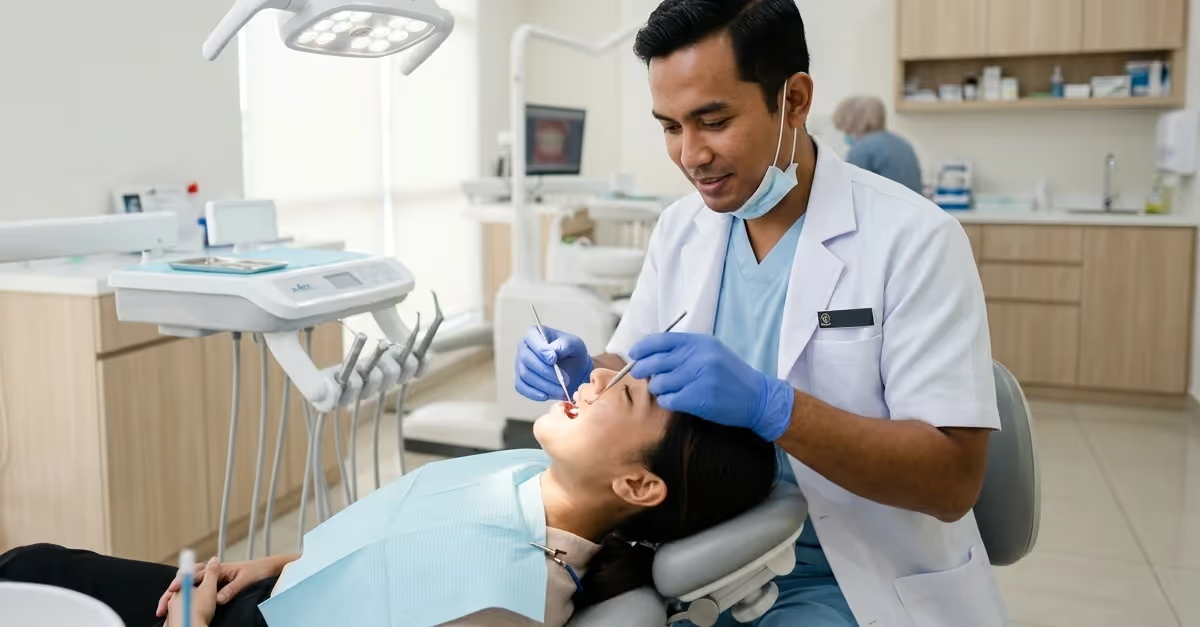
Enter Southeast Asia’s dental market with a clear strategy: navigate regulatory complexity, sequence market entry, and retain control of registrations to scale efficiently.
.avif)
Learn about Cisema’s redesigned website with faster performance, clearer navigation and expanded Asia Pacific coverage.

Learn about license validity and renewal rules for medical devices in Southeast Asia.

Explore fast-track medical device registration in Southeast Asia, cut timelines by 6–12 months, and accelerate market entry with Cisema.

Indonesia Enforces Annual Internal Audits for Medical Device Distributors
Indonesia’s Ministry of Health now enforces annual internal audits for medical device distributors under PP No. 28/2025.

Indonesia Expands Retail Access to Medical Devices
Indonesia has expanded retail access for selected low-risk medical devices, opening new sales channels to benefit consumers.

Indonesia Updates Registration Timelines for Medical Devices & IVDs
Indonesia revises medical device and IVD registration timelines under Permenkes 11/2025, extending approval timelines.

Indonesia MoH Announces Temporary Closure of Medical Device Regulatory Systems
Indonesia’s Ministry of Health will temporarily close key medical device licensing and registration systems from December 2025 to January 2026

Indonesia BPOM Strengthens Cosmetic Clinical Trial Requirements Under Regulation No. 34 of 2025
Indonesia’s BPOM has strengthened clinical trial approval procedures and introduced detailed requirements for cosmetic clinical trials.

BPOM Clarifies Indonesia–US Trade Agreement Impact on Pharmaceutical Regulation
BPOM confirms pharmaceutical authorization, GMP oversight, and regulatory authority remain unchanged under the Indonesia–US trade agreement.

Indonesia Revises KBLI Classification Structure Affecting Regulated Wholesale Activities
Indonesia’s BPS has updated KBLI business classification codes, impacting licensing, OSS data, and compliance for regulated sectors.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
