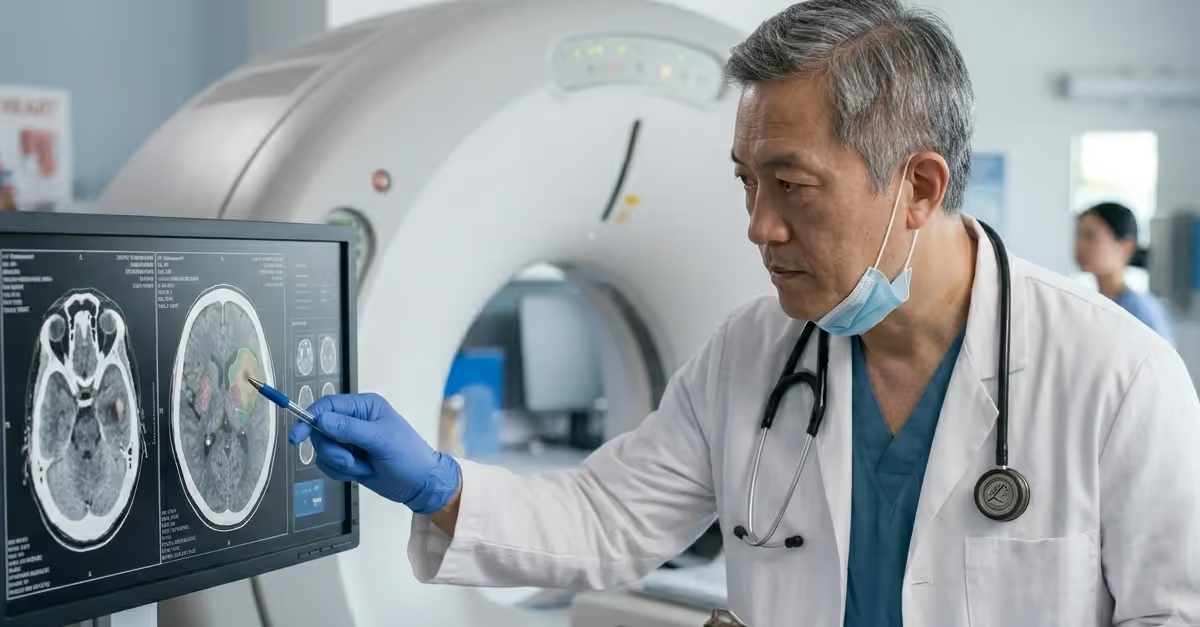
China Type Testing Medical Devices & IVDs
Type testing, also called in country testing, is a critical step in the regulatory process for medical devices and IVDs seeking approval in China. When conducted in accordance with national standards set by the National Medical Products Administration (NMPA), type testing verifies that products meets performance, safety, and quality requirements before they can be legally marketed.
This process ensures that products are safe for patients and comply with China’s stringent regulatory framework. The final testing report is an essential document reviewed by authorities for product approval.


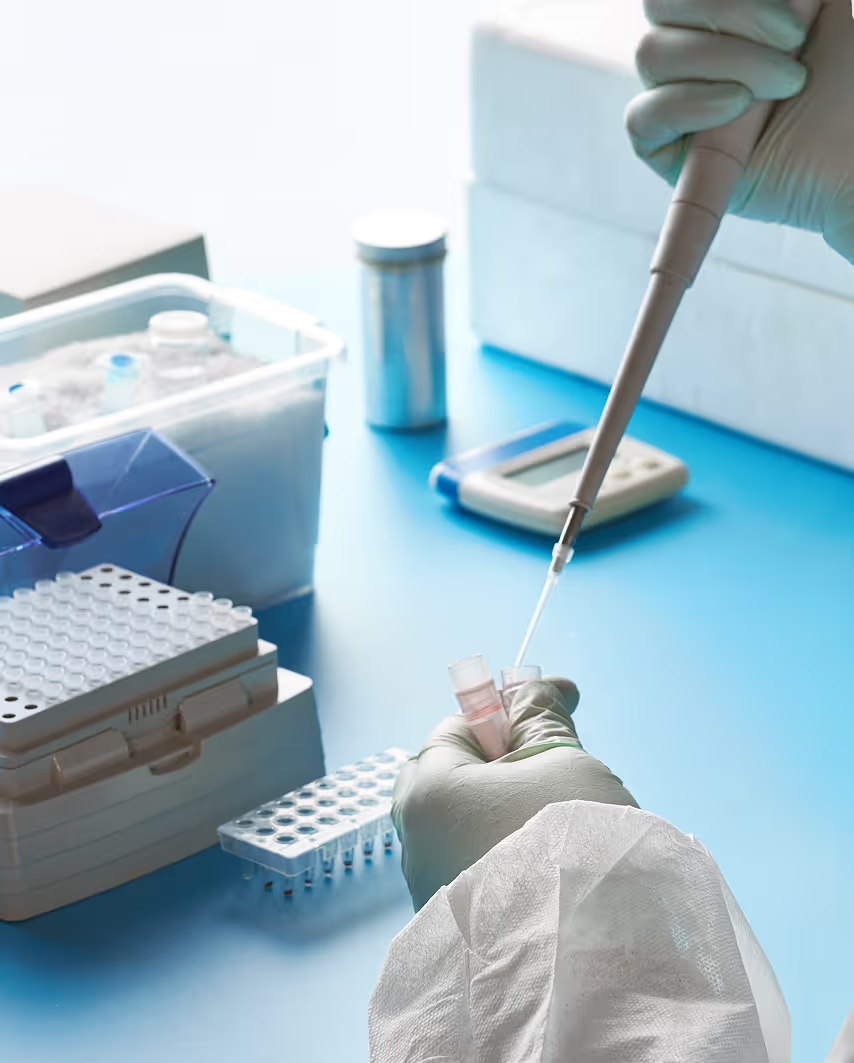
IVD & Medical Device Testing
Medical devices and IVDs must be tested at an NMPA accredited laboratory, where the product samples are tested against the applicable GB and YY standards. These tests check a variety of aspects including, but not limited to, electrical safety (especially for electrical medical equipment), biocompatibility, mechanical performance, and electromagnetic compatibility.
The tests depend on the product’s intended use and classification. The specific test report from these tests forms an integral part of the required documents for the registration application with the NMPA, emphasizing the role of the testing report as a critical document in the registration application. This includes demonstrating conformity to the Product Technical Requirements specific to each product type.
It is also important to include certificates of critical components as part of the necessary documentation for ensuring safety and usability.
Regulatory changes in October 2021 allowed for self-testing under certain conditions, however the process remains complex and is primarily only feasible for large domestic manufacturers with the resources to generate to a self testing report in compliance with NMPA guidelines. For an overseas manufacturer, compliance is easier when working a certified NMPA laboratory, especially given language barriers and the evolving nature of China's testing requirements.
Due to these language issues, complex regulations, and stringent testing measures, many manufacturers hire local regulatory experts such as Cisema, who handle and oversee the type testing process for faster and more accurate results.
How Long Does Type Testing Take?
The duration of type testing in China typically ranges from 4 to 6 months under standard conditions. However, if biocompatibility testing is required, the process may extend to up to 9 months. The high demand for testing services can lead to long waiting times for type testing dates.
Several factors can influence the overall timeline, including:
Device complexity
Scope of testing
Laboratory workload
Documentation & sample submission
Responsiveness to laboratory questions
Biocompatibility tests
Our China Type Testing Services for Medical Devices & IVDs
We provide end-to-end support for Class ii and Class iii medical device and IVD type testing in NMPA-accredited laboratories. Local testing institutes play a crucial role in the evaluation of products, and their involvement is essential to ensure thorough and accurate testing processes. To encounter any difficult testing scenarios, we send our experienced test engineers on-site to supervise all testing in China to guarantee accurate and reliable results.
Our services include:
Pre-Testing Consultation
Coordination with National Medical Products Administration (NMPA)-Accredited Laboratories
On-Site Supervision by Test Engineers
Our test engineers are present on-site to oversee the entire testing process. They ensure that all testing details are meticulously addressed during the type testing procedures, facilitating comprehensive evaluations. This includes interactions with reviewers to confirm that all protocols are followed correctly.
Test Progress Tracking & Testing Report
Interpretation of Test Results
Documentation & Certification Support
Post-Testing Advisory
Request a Proposal from Our Life Sciences Team for Type Testing Your Medical Device or IVD in China
Let Cisema help turn your plans into reality.
Explore Related Services
Whether you’re just starting out or are a blue-chip, access our wide range of services and solutions to help your business thrive.
China Medical Device Registration
We guide you through every step of Class i filings and Class ii and iii registrations. We take care of compliance with NMPA requirements, advising you on the optimal market entry pathway, and helping you through the whole process.
China NMPA IVD Registration
We support you through every stage of the China IVD registration and filing process, ensuring compliance with NMPA regulations. From choosing the best market entry strategy to handling regulatory requirements, we make the process as smooth as possible.
NMPA Legal Agent
We are your authorized representative in China, and fulfill your registrations and filings as well as ensuring compliance with quality and PMS requirements.
NMPA Clinical Evaluation Report
Clinical Evaluation Reports can avoid the need for a China clinical trial in some instances. Our experts can advise and prepare NMPA compliant CERs.
Drug Device Combination Products
We assist with the classification, registration, and compliance of combination products in China, whether drug or device led.
China Clinical Trials
You may be required to provide supplementary clinical data on Chinese patients or conduct a full clinical trial in China.Our clinical research team will advise you, prepare the study protocol and assist setting up the clinical trial in China.
Medical Device PMS & Vigilance
The NMPA is placing greater focus on Post-Market Surveillance (PMS) such as more in-market sampling inspections and regular standard changes.
Regulatory Outsourcing
If you are seeking specific specialist expertise or have ongoing resource-intensive needs, we have a variety of solutions to meet your needs.
China Medical Device Quality Compliance
We help you achieve and ensure ongoing compliance with Chinese requirements.Our experts prepare and assist you for official NMPA inspections.






%20Market%20A%20Guide%20for%20Foreign%20Manufacturers%20(1).avif)