News
Coronavirus product manual changes are required by the NMPA in China. Notice No. 349 (2023) outlines important modifications for Coronavirus nucleic acids, antigens, and antibodies detection reagents.

Avoid animal testing for general cosmetics and special cosmetics in China will be the main topic of this Spanish webinar

China health food raw material update has been issued by the State Administration for Market Regulation, along with the NHC and the NATCM.

China prohibited cosmetic ingredient list update has been proposed by the National Institute for Food and Drug Control (NIFDC).
China medical device standards update plan 2023 has been announced by the National Medical Products Administration (NMPA).
IVD Reagent Registration Change Review Draft Guidelines have been issued by CMDE in draft format for industry comment by June 30, 2023.
China suspends bone graft material from HansBiomedCorp (South Korea) following a remote off-site inspection by the NMPA.

Physicochemical characterization of nanomaterials used in medical devices encompasses the evaluation of various properties

Registration of disposable light-proof infusion sets in China shall refer to the technical review guidelines revised by the CMDE in April 2023

Registration of disposable blood separation devices in China shall refer to the technical review guidelines revised by the CMDE in April 2023.

Registration of disposable infusion devices in China shall refer to the technical review guidelines revised by the CMDE on April 28, 2023.

Registration of blood dialyzers in China shall refer to the technical review guidelines revised by the CMDE on April 28, 2023.

Registration of Blood Dialysis Concentrates in China shall refer to the technical review guidelines revised by the CMDE on April 28, 2023.
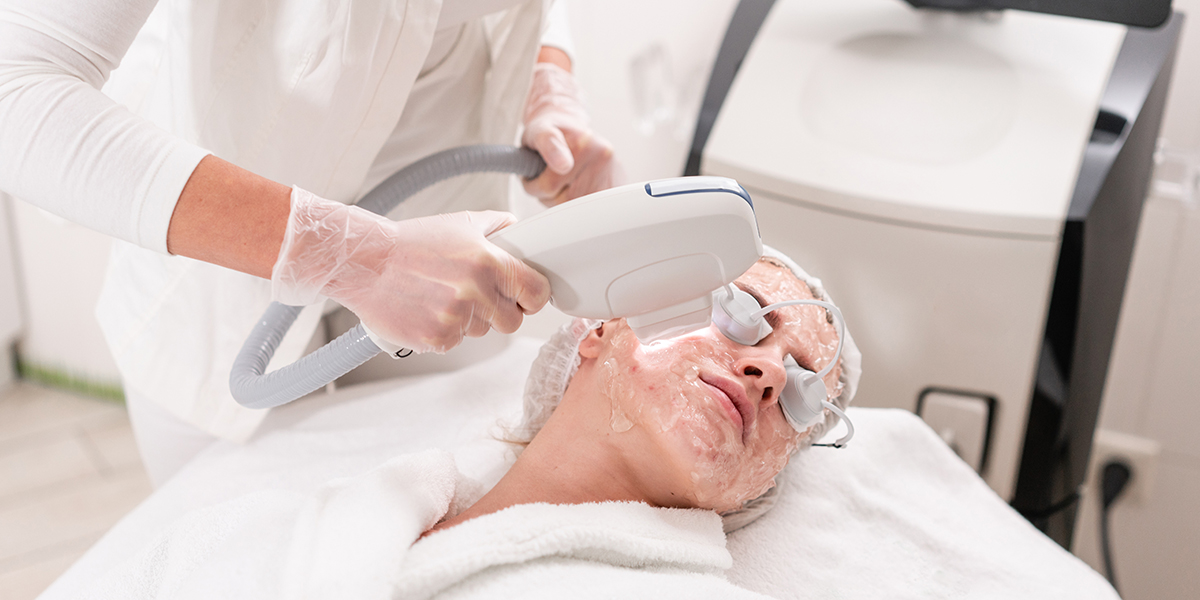
Intense pulsed light therapy device registration in China shall refer to the newly revised technical review guidelines issued in April 2023

China motorcycle rearview mirror and helmet standard requirements - GB 15084-2022 and GB 811-2022 will come into effect on July 1, 2023.

Clinical trials of sodium hyaluronate complex solution have a set of draft guidelines issued by the China CMDE on May 17, 2023.
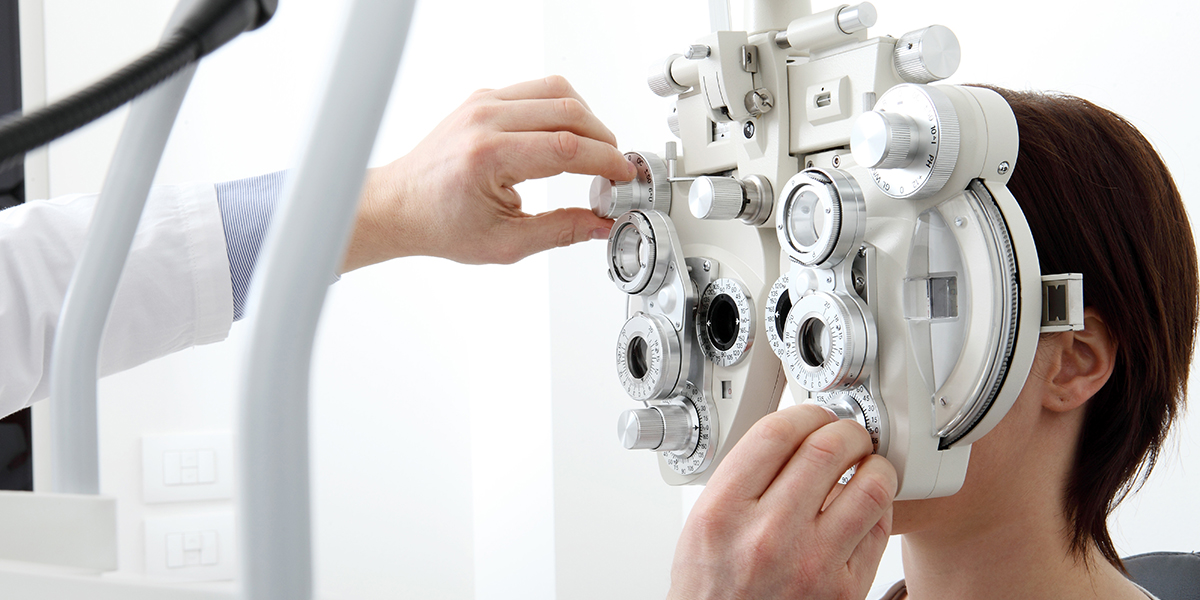
China registration of ophthalmic optical measurement devices shall refer to the respective technical review guidelines issued by the CMDE

Pre-communication for Hainan medical device clinical RWD applications will be required to follow the CMDE trial measures.

The registration platform for cosmetic raw materials safety information in China helps ensure that businesses fulfill their responsibilit...

Technical guidelines for reporting cosmetics formulas were drafted by the National Institutes for Food and Drug Control.
Get in Touch with Our Consultants
And discover how we can support you.
