News

China medical device quality safety responsibility regulations were issued by NMPA and will come into force on March 1, 2023.

China drug standards draft measures help ensure the safety, effectiveness and quality controllability of drugs in China

China special equipment safety supervision regulations are undergoing review by the State Administration for Market Regulation (SAMR).

China special equipment quality and safety responsibilities for production entities have draft provisions released by the SAMR.

Cisema 2023 events cover seminars, webinars, trainings, etc., to help manufacturers stay ahead of China regulatory developments

Cosmetics enterprises quality and safety responsibilities have been addressed in the draft regulation issued by the NMPA on Nov. 29, 2022.

Cosmetics adverse reactions collection and reporting draft guidelines for registrants and filers (trial) are formulated according to the CSAR

China cosmetics filing FAQs is a publication first issued by the Beijing Municipal Medical Products Administration (MPA) in November 2021.
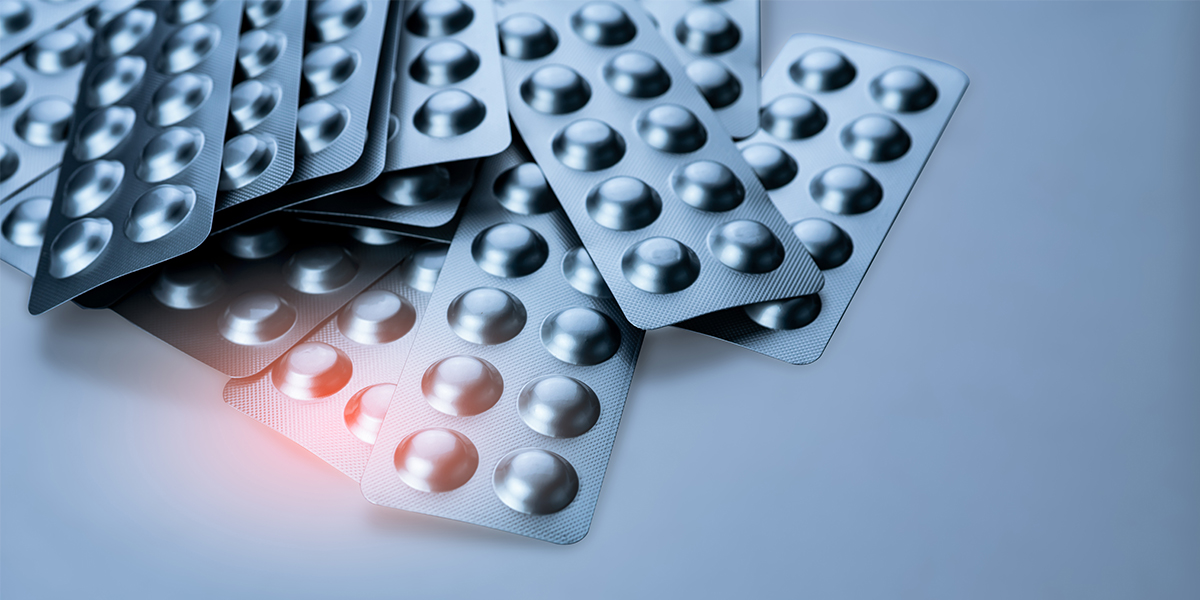
Drug recalls in China now designate market authorization holders as the key responsible entity, and have a revised regulatory framework.

Electronic submission for China drug registration applications will be available from January 1, 2023, onwards.

China post-market surveillance regulations for medical devices in 2022 were being reviewed in a meeting carried out by the NMPA.

China UDI requirements will cover various class II medical devices manufactured after June 1, 2024, once the regulation comes into effect.

Regulation on safety supervision of special equipment in China had a new draft amendment issued by the China SAMR on November 30, 2022.

China special equipment production unit responsibilities for quality and safety shall be fulfilled once the SAMR provision is in effect

China medical device quality and safety responsibilities have seen new proposals issued by the NMPA recently

China food production licensing rules and principles have been updated by the SAMR and took effect on November 1, 2022.

China's digital health regulatory framework for SaMD has been evolving with several key legislations implemented over the years by the NMPA.

Changes during China drug registration shall refer to the working procedures issued by the Center for Drug Evaluation on November 11, 2022.

China medical device registration electronic certificates have the same legal effect as the paper version of the registration certificate.

China IVD Reagent Registration and Review Guidelines have been recently updated. Six guidelines came into effect on September 28, 2022.
Get in Touch with Our Consultants
And discover how we can support you.
