IVDs
China radio frequency beauty devices have new technical review guidelines issued to ensure that products are safe and effective

China medical device industry standards March 2023 update covers 20 standards released by the NMPA for manufacturers to comply with.
China’s IVD reagent classification catalogue has been revised and reissued for industry comment. The deadline for submission is Apr. 12, 2023

China IVD Reagent Registration and Review Guidelines have been recently updated. Six guidelines came into effect on September 28, 2022.

The China Clinical Trials GCP (Good Clinical Practice) 2022 amended version will come into effect on May 1, 2022.
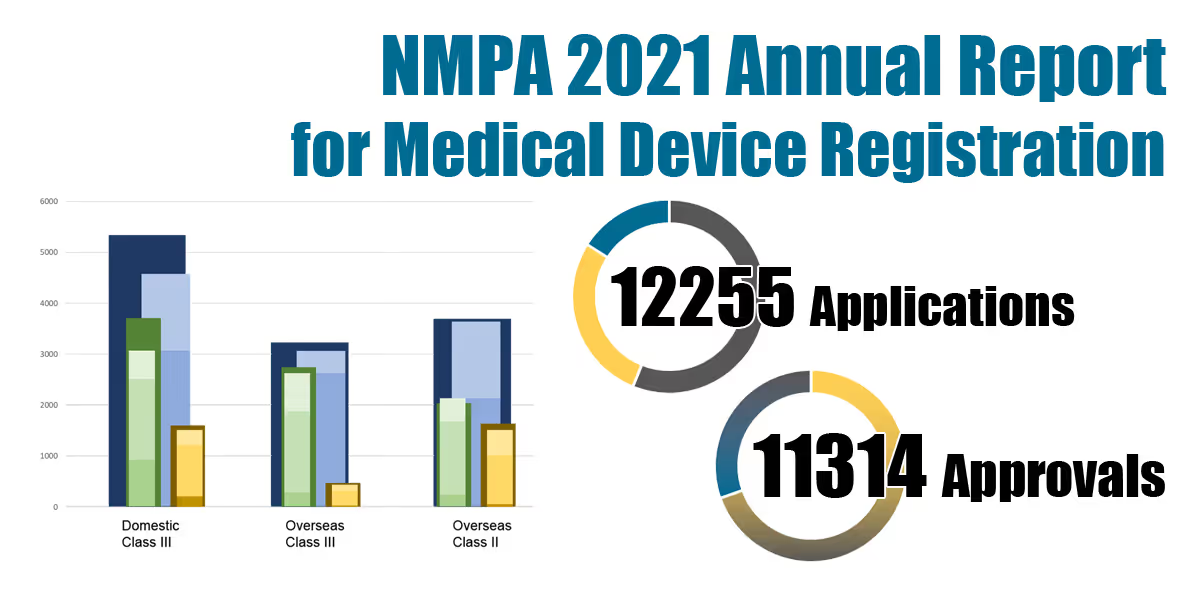
The NMPA reported a total number of 12,255 applications and 11,314 approvals for medical device and IVD registrations in 2021.

Overseas clinical trial data for IVD registration in China now adopts the new technical guidelines issued on December 1, 2021, by the NMPA.
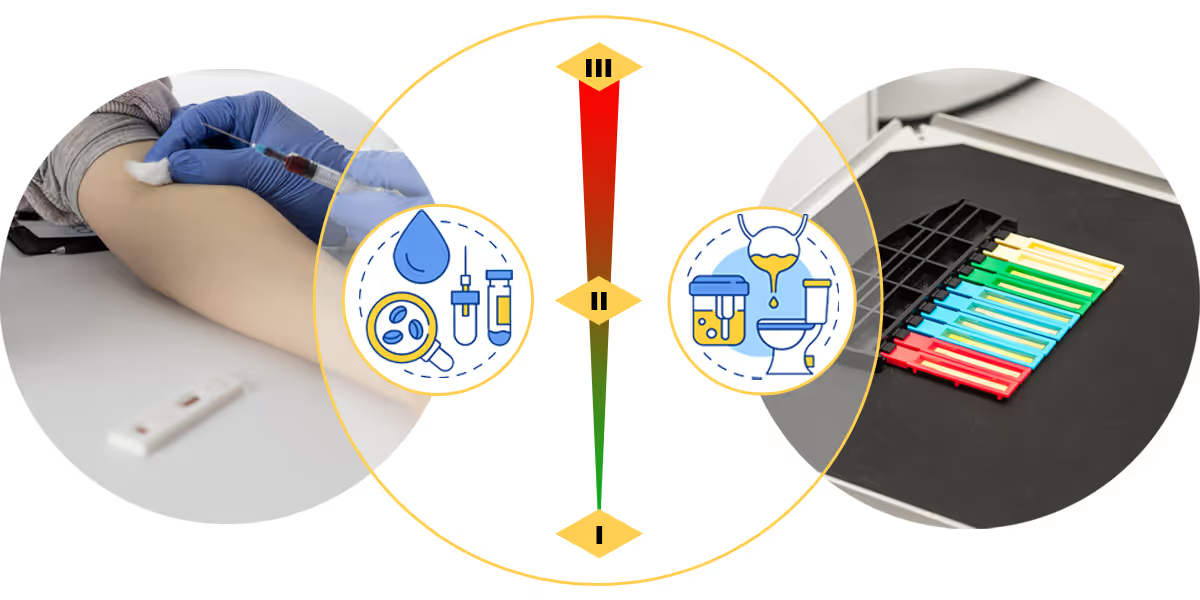
On October 29, 2021, the NMPA published the In Vitro Diagnostic Reagents Classification Rules for the first time as an individual document.

NMPA published a revised the clinical trial exemption list for medical devices and IVD reagents which became effective on October 1, 2021.

The NMPA announced the plan for the implementation of UDI for the remaining class III products which do not yet require UDI.

The new medical device (Order 47) and the new IVD (Order 48) regulations will be effective from October 1, 2021 onwards.

The CMDE published several interesting case studies to address some of the detailed requirements for IVD and SiMD registration in China
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
