IVDs

NMPA published a regulatory draft to require all class III medical devices and IVDs manufactured after March 1, 2022 to have UDI.

New revised law on the Supervision and Administration of Medical Devices (Order 739) issued by the State Council came into force in June 2021

The NMPA is currently soliciting public comments on the draft documents. The public consultation period is open until May 30, 2021.
6 technical guidelines & 4 GB / YY draft standards were announced; 2 products registered & 7 applications accepted by the fast-track approval pathway.
On January 19, the NMPA announced the new clinical trial exemption list for medical devices and in-vitro diagnostics reagents.
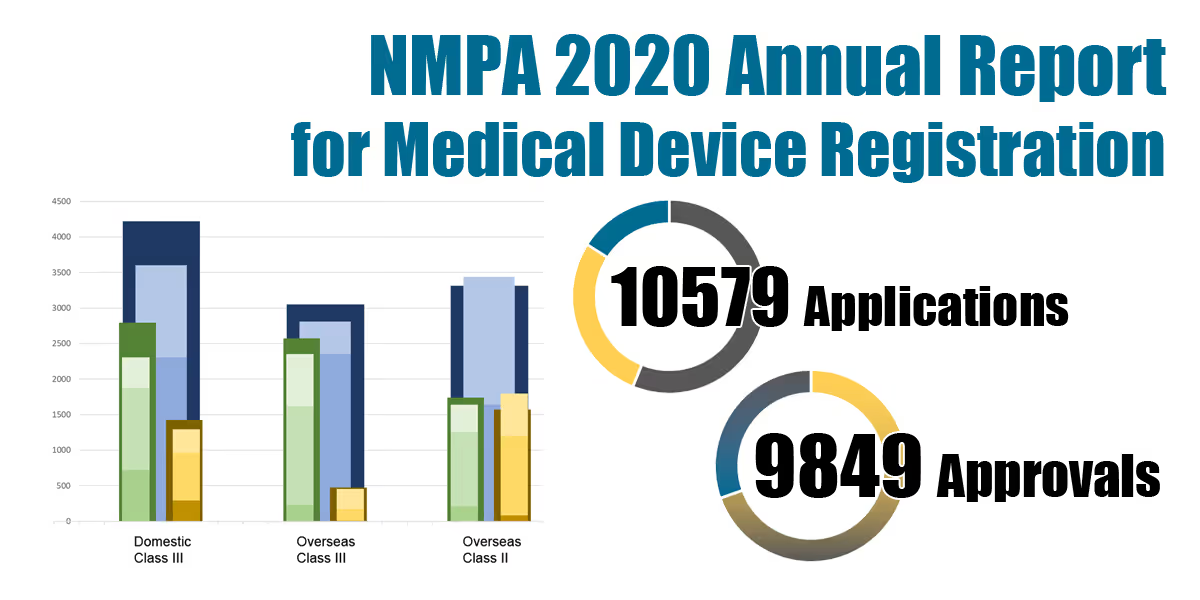
A total number of 10579 applications and 9849 approvals for medical device registrations in China were reported in 2020 along with a detailed breakdown.

NMPA’s recent release of the updated product information of 29 IVD reagents, primarily to down-classify from class III to class II, was long overdue.

On July 29, 2020 the CMDE (Center for Medical Device Evaluation) of NMPA (National Medical Products Administration) released a draft of the proposed amendment to the list of medical devices and IVDs to be exempted from clinical trials:
The draft proposes to add 53 medical devices (Class II: 34; Class III: 19) and 15 IVDs (Class II: 13; Class III: 2) to the clinical trial exemption list. Also, the names and descriptions of 11 medical devices (Class II: 9; Class III: 2) are proposed for revision, while there are 2 medical devices being combined with other products.
Here are the links to:
Proposed amendment for the list of medical devices to be exempted from clinical trials
Proposed amendment for the list of IVDs to be exempted from clinical trials
The CMDE is currently soliciting for public comments, and the public consultation ends on August 31, 2020.
By Jacky Li. Contact Cisema if you would like to learn more.

On June 9, the CMDE (Center of Medical Device Evaluation) of NMPA (National Medical Products Administration) announced the initiative to draft the technical review guidelines for the registration of:
- Reagents for rotavirus antigen tests
- Real-time qPCR instruments
- Reagents for testing IgM and IgG antibodies against HPV B19
- Reagents for GBS nucleic acid tests
Manufacturers of the above products were invited to sign up and cooperate with the CMDE to provide any needed assistance.
On July 1, the CMDE released the technical review guidelines (2nd draft) for the change of licensing items for IVD Reagents. The CMDE is currently soliciting public comments until July 25, 2020.
On July 3, the CMDE of NMPA announced the initiative to draft the technical review guidelines for the registration of IVD reagents utilizing overseas clinical data.
By Jacky Li. Contact Cisema if you would like to learn more about the above news.

Cisema will jointly present a webinar session with the Chamber of Commerce (IHK) Lübeck on August 29 to provide an overview of the market potential and registration of medical devices and IVDs in China.
The session will be interactive, and will go through many interesting topics:
- The Chinese market
- The authorities and other players in China
- The admission requirements and responsibilities
- Legal representation of the manufacturer - Legal Agent
- Post Market Surveillance
- Recent changes in the registration process
- New approaches to simplified market access in certain regions
Date: August 26
Time: 9:00 am – 11:00 am
Price: Free of charge
Open for public
Our speaker: Markus Roepke
Language: German

NMPA solicites public opinions on the draft proposal to reclassify IVD reagents in the 6840 Classified Subdirectory (2013 Version).

On 20.12.2019, the China NMPA (National Medical Products Administration) announced (No.91-2019) that it would add 148 medical devices and 23 in-vitro diagnostic reagents to the clinical trial exemption list, as well as to revise the names and descriptions of 48 medical devices and 4 in-vitro diagnostic reagents. Medical devices and in-vitro diagnostic reagents on the clinical trial exemption list won’t require completion of clinical trials in order to achieve NMPA certification.This new clinical trial exemption list is effective since the date of announcement on 20th December.Feel free to contact us if you would like to know what products were included in the clinical trial exemption list for medical devices, and the clinical trial exemption list for IVDs.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
