Medical Devices

OEM quality agreement for medical devices helps to set out the the rights, obligations and responsibilities of registrants, filers and OEM...

Medical device clinical trial data submissions in China should now refer to the the NMPA technical review guidelines issued on Nov 25, 2021.
22 high-risk medical implants have been added in the draft catalogue of medical device products to be prohibited from China OEM production.
The NMPA issued a list of common violations committed by medical beauty service providers in China throughout 2020 and 2021.

Cisema presented on the topic of registration and approval of medical devices in China to 94 participants, and held a Q&A session after.
The NMPA published various technical guidelines covering specific topics on clinical trials, clinical evaluation, and animal testing.
Regulatory Measures for Self-testing finalized by National Medical Products Administration on October 22, 2022.
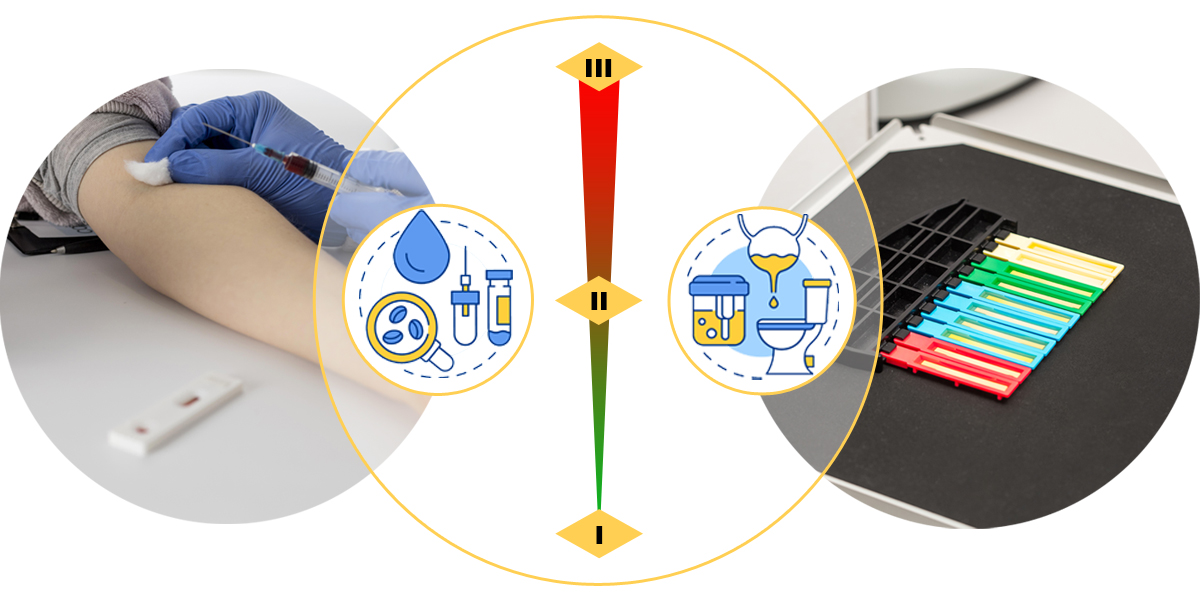
On October 29, 2021, the NMPA published the In Vitro Diagnostic Reagents Classification Rules for the first time as an individual document.
CMDE Drafted regulations for metallic implants using additive manufacturing and for dental implants and dentistry products
Technical review guideline for the clinical evaluation of Intravascular Catheters products in comparison with predicate devices was announced

On September 18, 2021, the China NMPA announced the implementation of a work plan to ensure product quality and safety for medical devices.

NMPA published a revised the clinical trial exemption list for medical devices and IVD reagents which became effective on October 1, 2021.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
