Medical Devices

The CMDE released the draft of technical review guidelines for submission requirements of medical device clinical trial data.

Drug release studies may be required for combination products in China that intend to release drug components to certain parts of the body.
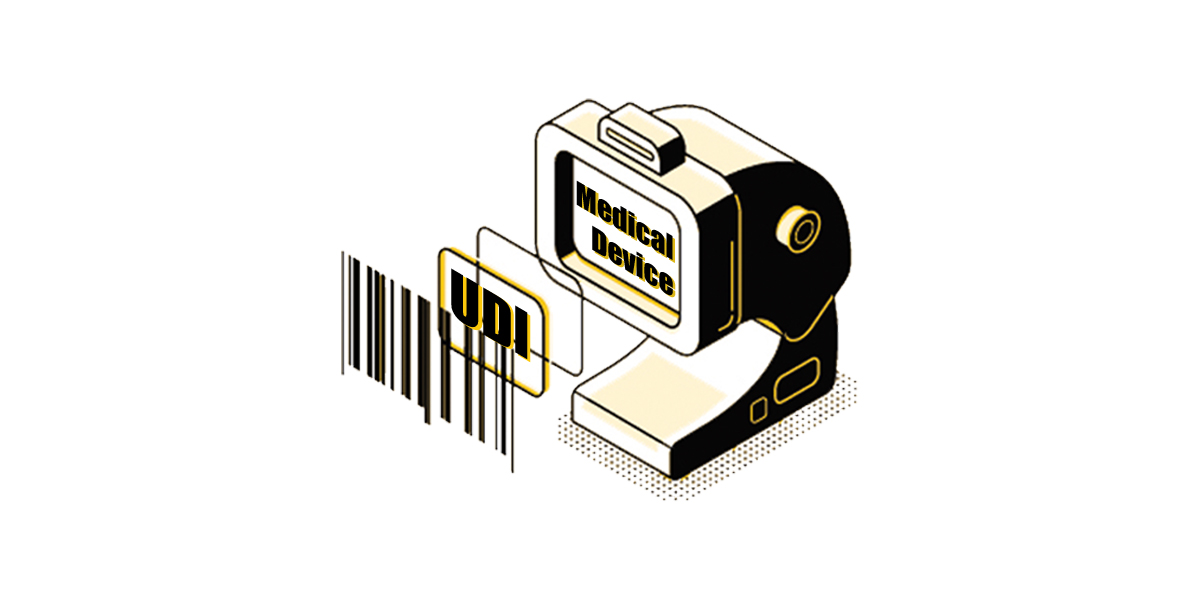
The NMPA announced the plan for the implementation of UDI for the remaining class III products which do not yet require UDI.
This is the first set of guidelines in China published specifically for nanomaterials used in medical devices in China.

The new medical device (Order 47) and the new IVD (Order 48) regulations will be effective from October 1, 2021 onwards.
NMPA published naming guidelines for Neurological and Cardiovascular Surgical Instruments & Medical Diagnostic and Monitoring Instruments

The CMDE published several interesting case studies to address some of the detailed requirements for IVD and SiMD registration in China

The NMPA is revising the Class I Medical Device Classification Catalogue to cover 600 new product examples.

China clarifies rules for registration and classification of drug-device combinations and read more about the NMPA

Due to stricter risk managements imposed by the EU MDR, there could be a high approval rate for EU medical devices registering in China.

The NMPA implemented the “Notice on Matters related to the Registration of Drug-Device Combination Products” (No.52-2021).

NMPA revised the draft “Required Working Measures on Self-Testing for the Registration of Medical Devices” to support Order 739.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
