Medical Devices

The draft amendment covers the guiding principles for the shelf-life of passive implantable medical device for registration and filing.
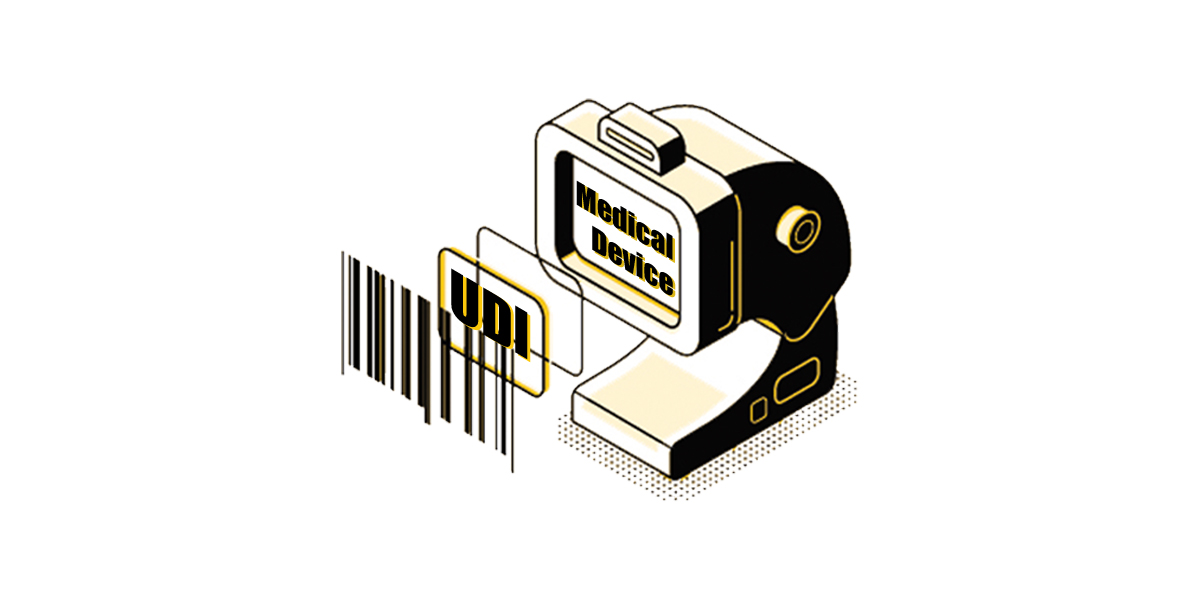
NMPA published a regulatory draft to require all class III medical devices and IVDs manufactured after March 1, 2022 to have UDI.

NIFDC announced 5 drug-based combination products and 5 medical device-based combination products among a total of 22 application results.

On July 8, 2021, the China National Medical Products Administration announced the classification guidelines for AI medical devices and SaMDs.
Popular beauty devices are classified as medical devices according to the classification catalogue in China
Biocompatibility testing is the one test that can be done outside of China provided it complies with the GB/T 16886 and ISO 10993 standards.
The NMPA (National Medical Products Administration) issued the Catalogue of Technical Guidelines for Reviewing Medical Device Registrations.

The catalogue covers 1,791 medical industry standards that are currently in effect. 77 industry standards will be revised this year.

List of institutions qualified for conducting clinical trials and also completed the filing application to be listed in the NMPA database.

New revised law on the Supervision and Administration of Medical Devices (Order 739) issued by the State Council came into force in June 2021

5 new technical medical device guidelines whilst there were 4 guidelines implemented and 6 drafts are announced by NMPA

The Center for Medical Device Standardization Administration issued the notice to solicitate proposals for amending medical device standards in China
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
