Medical Devices

The NMPA is currently soliciting public comments on the draft documents. The public consultation period is open until May 30, 2021.

883 commodities were announced under the MFN Rate of Duty whilst the import tariff rate on artificial heart valve, hearing aids, etc., reduced from 4% to 1%
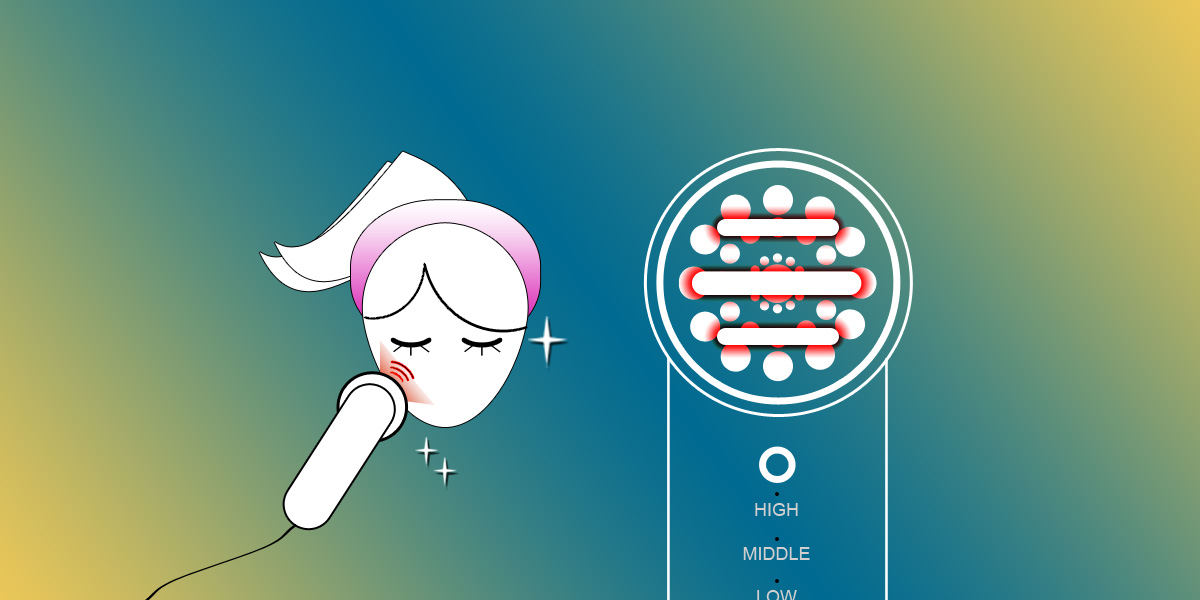
The China NIFDC announced the technical guidelines (draft) for Radio Frequency beauty devices to be regulated as class II or class III medical devices.
Our testing engineers are specialized in EMC (Electro Magnetic Compatibility), electrical safety, physical and chemical property and biocompatibility tests.

9 product categories have been reported by the NMPA for not meeting the standard requirements whilst 12 regulatory guidelines and drafts have been issued.

According to the announcement published by the Centre for Medical Device Evaluation) of the National Medical Products Administration on April 12, 2021...
The announcement cover details of the working procedures and appendices for quality spot checks and inspection procedures for medical devices within China.

Inspection work shall be carried out in accordance with the mandatory standards or product technical requirements for post-market activities in China.
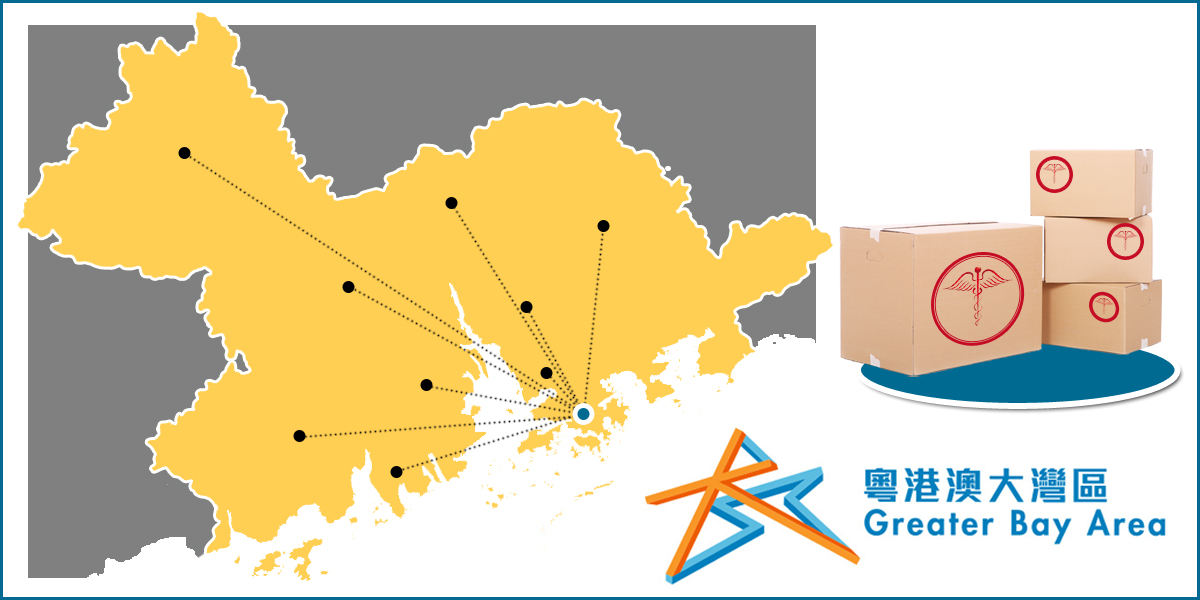
The Guangdong MPA announced the first approved medical device arriving the University of Hong Kong – Shenzhen Hospital via the Greater Bay Area initiative.

22 technical guidelines (draft) were issued in March 2021, and technical guidelines for products monitoring non-invasive blood / glucose will be drafted

The NHC announced the implementation of The Administrative Measures for the Clinical Use of Medical Devices coming into force from March 1, 2021 onwards.

The Guangdong MPA (Medical Products Administration) announced details about the pilot plan and the responsibilities of different departments involved.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
