Medical Devices

The NMPA announced the implementation of a mNMPA Introduces the Medical Device Master Filing System covering both medical devices and IVDs.

The NMPA announced a total of 28 updates on the classification catalogue for medical devices including 15 classification changes and 13 content updates.
6 technical guidelines & 4 GB / YY draft standards were announced; 2 products registered & 7 applications accepted by the fast-track approval pathway.

On February 5, the NMPA issued a report covering a breakdown of the total existing and new standards for medical devices in 2020.

On January 19, the NMPA announced the new clinical trial exemption list for medical devices and in-vitro diagnostics reagents.
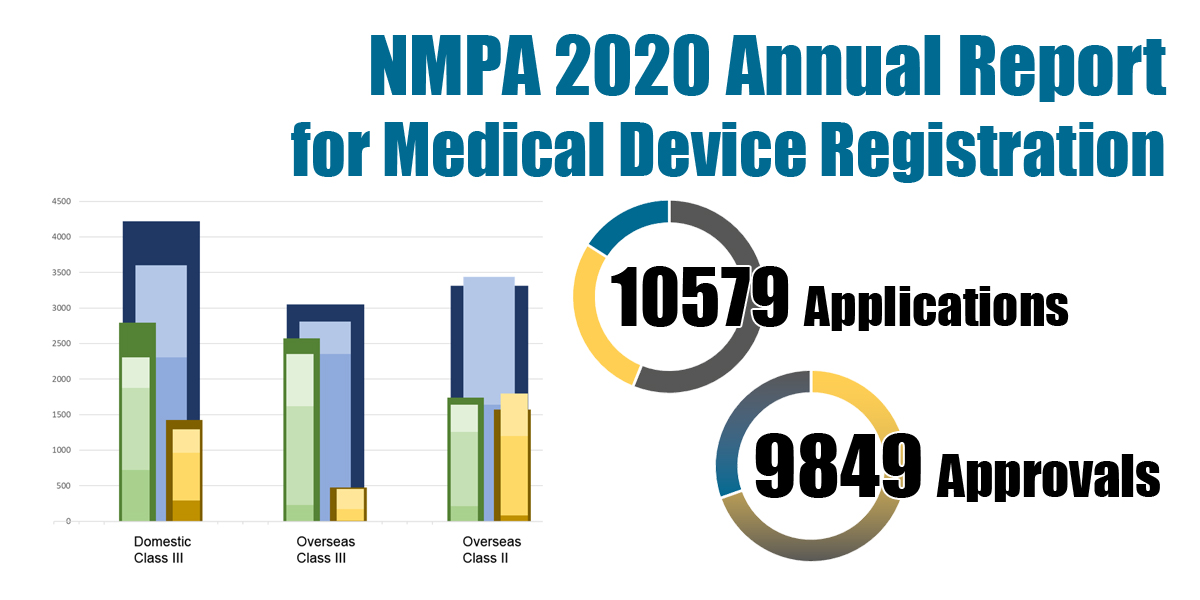
A total number of 10579 applications and 9849 approvals for medical device registrations in China were reported in 2020 along with a detailed breakdown.

Ultrasonic coupling agents are reminded to comply with mandatory standards and any necessary filing changes must be completed by the end of February 2021.
The new draft clarifies the registration requirements and categorization method of different drug-device combination products in China.

3 GB standards announced; 14 technical guidelines and 2 draft guidelines were issued; 6 medical devices pending for fast-track approval

Due to the 2020 performance of the fast-track approval process for medical device registration in China, the CDME plans to implement a smart review process.

The NMPA issued guidelines to address the standard procedure of submitting risk evaluation reports after the occurrence of adverse events.

The CMDE shared a timeline of events for establishing regulatory control on AI medical devices and shared the latest status of their upcoming plans.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
