Medical Devices

The CMDE is drafting pre-market evaluation guidelines for AI software in diagnostic pathology to support China’s upcoming 5-year plan

International Medical Device Regulator Forum Accepts China’s Proposal on “Post-Market Clinical Follow-Up Studies” and Seeks Industry Comment

In September 2020, the NMPA accepted 2 overseas applications from CooperVision and BIOMERIEUX for prioritized review and approval.

In September 2020, the NMPA issued technical review guidelines for medical devices including 3D-printed lower jaw prosthesis, customized bone implants, etc.

On September 14, the CMDE announced two new additions in the list of class III medical devices requiring clinical trials in China.

The NMPA updated the first batch of medical devices requiring UDI, and extended the UDID’s pilot phase to January 1, 2021
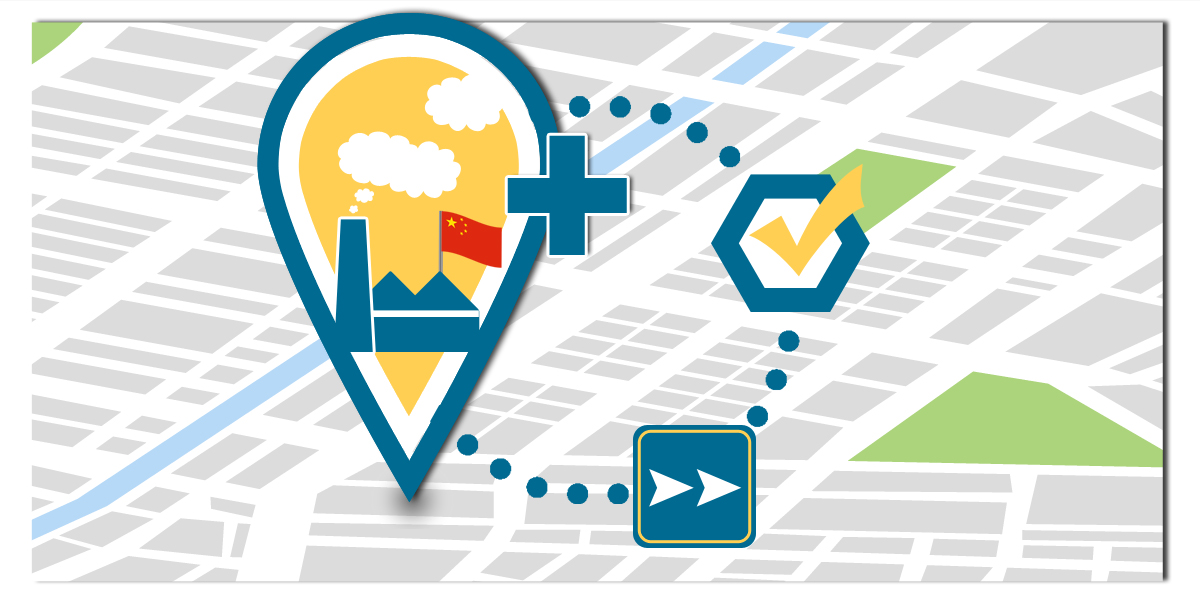
The NMPA optimizes the approval process for overseas manufacturers intending to produce their already registered class II or III medical devices in China.
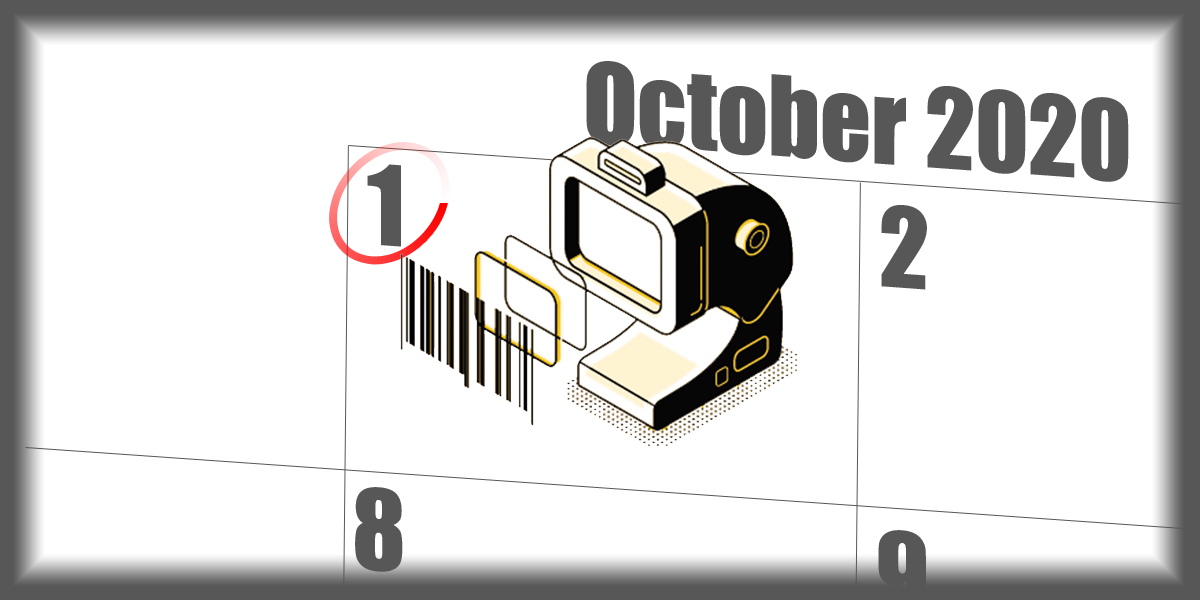
The NMPA released an introductory video about the online UDI database, soon to be implemented for the first batch of medical devices on October 1, 2020.

The China's State Council announced the regulatory framework for AI, and the plan to implement standards for AI developments in the healthcare industry.

Guidelines covering technical review / clinical trials / clinical evaluation for the registration of medical devices in China

2 medical devices were passed for the fast-track approval, whilst the NMPA accepted the applications for the fast track approval of 8 medical devices.
NMPA Issues 2019 Annual Report, Strengthening Scheme and Updates on Medical Device Mandatory Standards in China
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
