News

China is making it easier to transfer registrations of drugs initially manufactured abroad to Made-in-China status to improve availability and accessibility to medicines.

China’s NMPA is introducing new supervision measures for the medical device entrusted production. This will come into effect on June 1, 2024.

Cosmetic Adverse Event Reporting guidelines for China have been released by the National Center for Drug Adverse Event Monitoring.

Regulatory Science for Pharmaceutical Innovation and Internationalization Conference took place on April 28, 2024, sharing GBA market insights

China CCAP Electric Vehicle Supply Equipment (CCAP-GZ-464230:2024) revised rules released by CCAP on March 27, 2024.

Measures to optimize cosmetic safety assessment management in China have been released by NMPA and come into effect on May 1, 2024.
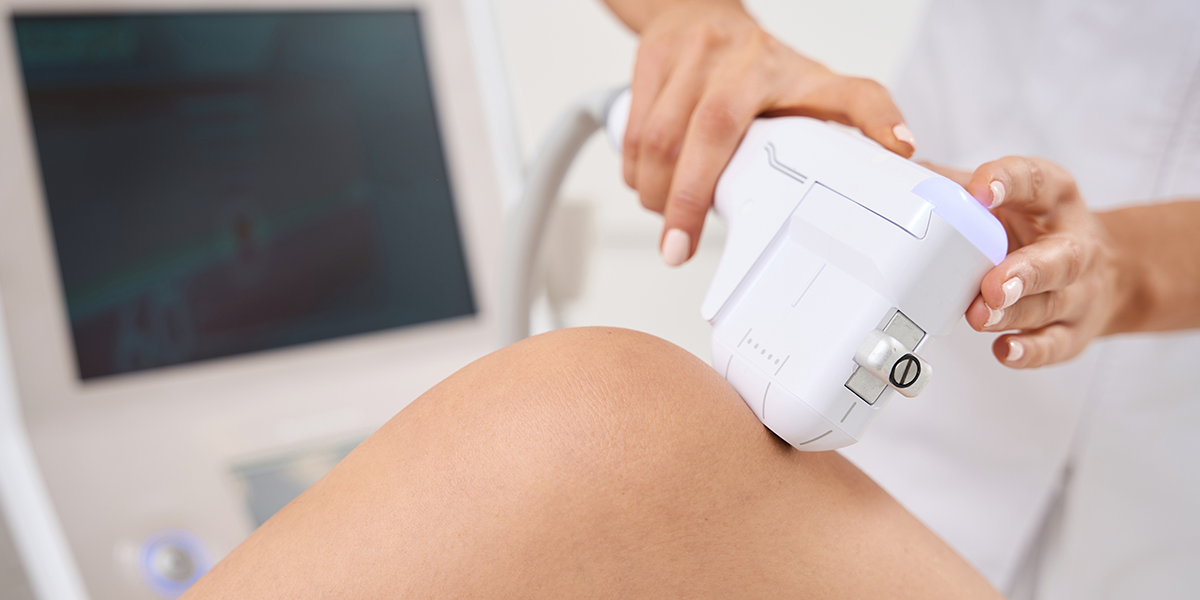
China RF Therapy Device classification clarification has been published by the NMPA. This notice is subject to medical device legislation.

Usability engineering for medical devices registration in China Guidelines were released by the CMDE on March 19, 2024.

GB9706.1-2020 deadlines updated and released by NMPA for compliance, medical device manufacturers must comply with GB9706.1-202 standards.

China gas appliance CCC certification and other products have been announced and implemented by the SAMR on April 7, 2024.

China cosmetics stability evaluation and other 3 technical guidelines have been drafted for public comments by the NIFDC on April 10,2024.

China’s NMPA has released its medical device sampling inspection plan for 2024. Find out if your medical device is due for inspection this year by reading our article.

China’s NMPA released 15 toothpaste testing methods on March 21, 2024. These testing methods are required for the notification all toothpaste products in China from December 1, 2024.

China’s NIFDC recently issued five technical guidelines on China cosmetic ingredients’ acute inhalation toxicity test ranging from experimental research to 28-day repeated dose tests.

China’s NMPA has recently released an update to the recommended clinical evaluation pathways for medical devices that are listed in the China Medical Device Classification Catalogue.

Hong Kong MDACS listing now accepts medical devices approved by Singapore HSA, with online submissions available starting April 15, 2024

China’s Center for Drug Evaluation has released its annual report for 2023.

China customs clearance requirements, with a focus on CCC and trade logistics, will be covered in the upcoming webinar on June 25, 2024

China’s SAMR has issued amendments to Special Equipment Production and Filling Licensing Regulations. They come into effect on June 1, 2024.

NMPA overseas factory inspection results have seen at least three medical device manufacturers notified by the authorities this week alone...
Get in Touch with Our Consultants
And discover how we can support you.
