News

The China CDE has issued its 2023 Annual Report reviewing the trend in Clinical Trials Applications for New Drug Registrations).

Drug registration in China for transferring Biological Therapeutics products marketed abroad to in China has a clear set of guidelines.

China Car Parts Certification will be issued by China’s Certification Alliance for Vehicles (CAV) for some car parts as of July 1, 2024. The certification process remains the same, however, the issuing body has changed to the CAV.

China compliant cosmetic ingredients now have a set of draft guidelines on how to determine and research their history of safe use.

China compliant cosmetic ingredients now have a set of draft guidelines on how to determine and research their history of safe consumption. They were released by China’s NIFDC on May 31, 2024.

China’s NMPA introduces Measures for the Supervision and Inspection of Medical Device CROs. They come into effect on October 1, 2024.

China Reference Listed Drugs (RLD) application requirements for Chemical Generic Drugs was issued by the CDE in 2022.

China motor vehicles CCC Certification and Implementation Requirements (CCAP-C11-01:2020) have been revised and published by CCAP on April 30, 2024.

China Compulsory Certification rules for Wire, Cable and other products have been revised and were published by CNCA on April 15, 2024.

China Contract Drug Manufacturing Provisions were issued back in October 2023 to strengthen the supervision and management of the entrusted production of drugs.

Formula food for special medical purposes (FSMP) clinical trial standards in China were recently updated by the SAMR on May 11, 2024.

China Cosmetic Adverse Event Reporting is crucial to maintaining compliance post-notification or post-registration of your cosmetics.

To improve the review process, a 60-day response period allows applicants to address any deficiencies identified by the Hong Kong Drug Office

China’s NIFDC issued guidelines for the submission of cosmetics safety assessment information on April 30, 2024.

China’s NMPA has released the rules concerning Cosmetics Inspections in China. They come into effect on November 1, 2024.

China low voltage components CCC certification reinstated by the CQC and came into effect on May 1, 2024.

China Circuit Breakers for Equipment standard GB/T 17701-2023 has been released by SAMR and came into effect from April 2024.
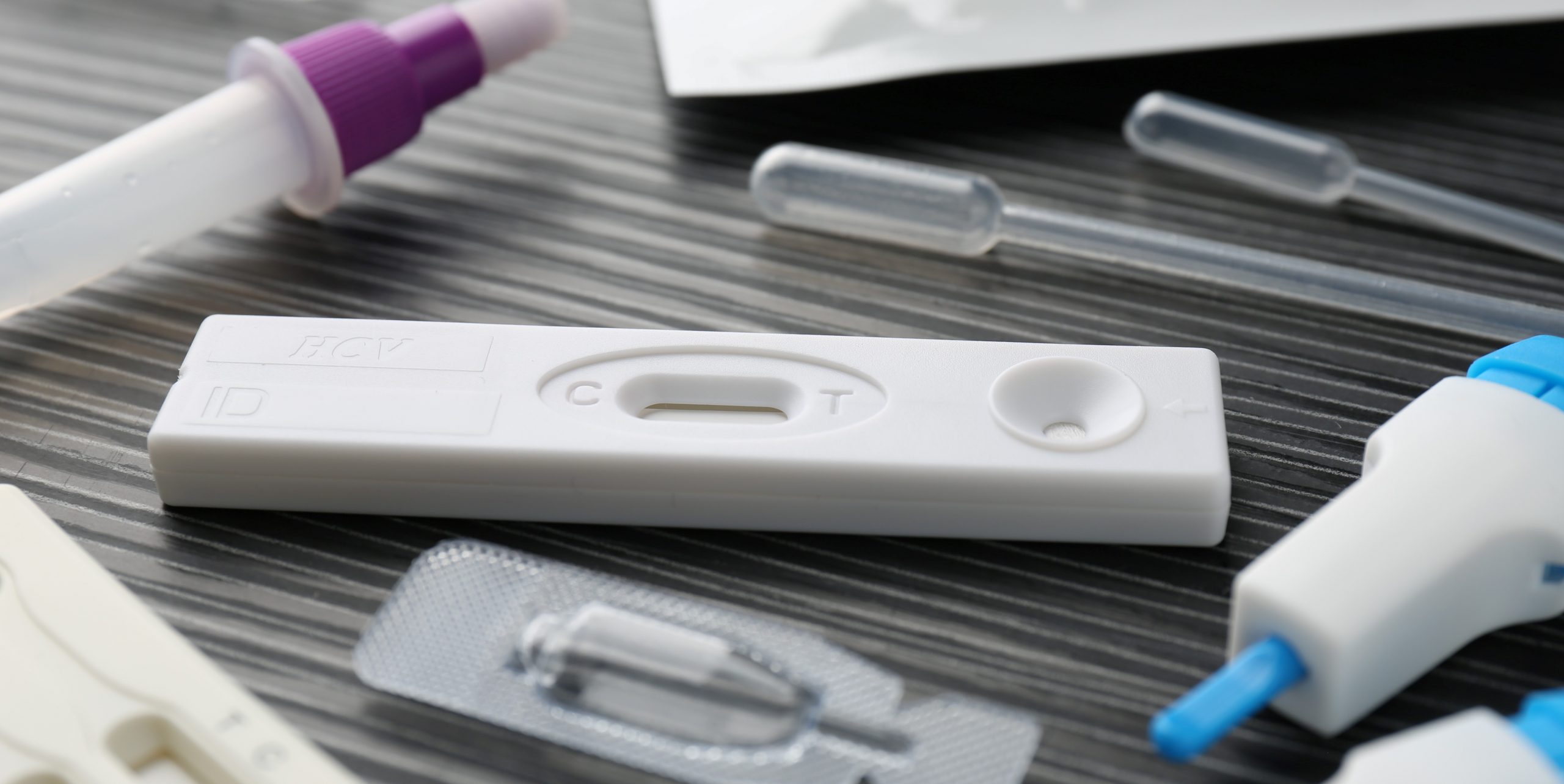
China has released an IVD reagent classification catalogue. It was issued by the NMPA and includes all IVDS except those used for blood screening and those labeled with radionuclides.

China veterinary drug comparative test requirements have been revised by the MOA and this regulation will come into effect from December 1, 2024.

China’s SAMR issued technical requirements for the notification of Ginseng, American Ginseng, and Ganoderma lucidum.
Get in Touch with Our Consultants
And discover how we can support you.
