News

China’s NMPA has suspended the import, sale and use of Cefodizime Sodium for injection by DAEWOONG BIO Inc following its rejection of an NMPA inspection request at its production site.
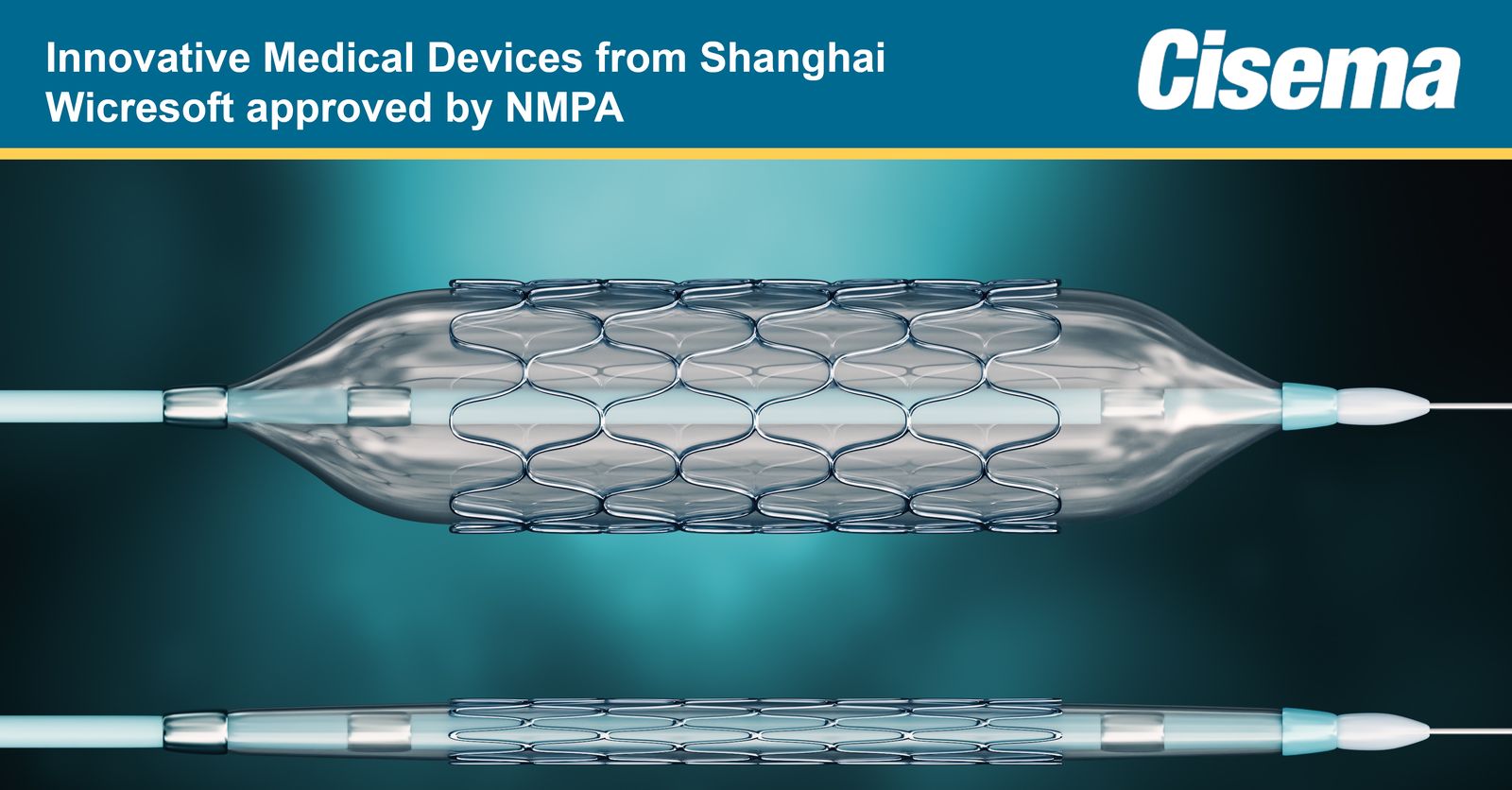
China’s NMPA has approved Shanghai Wicresoft’s Bioabsorbable Rapamycin Eluting Coronary Stent System through the Innovative Medical Device Green Pathway Programme.
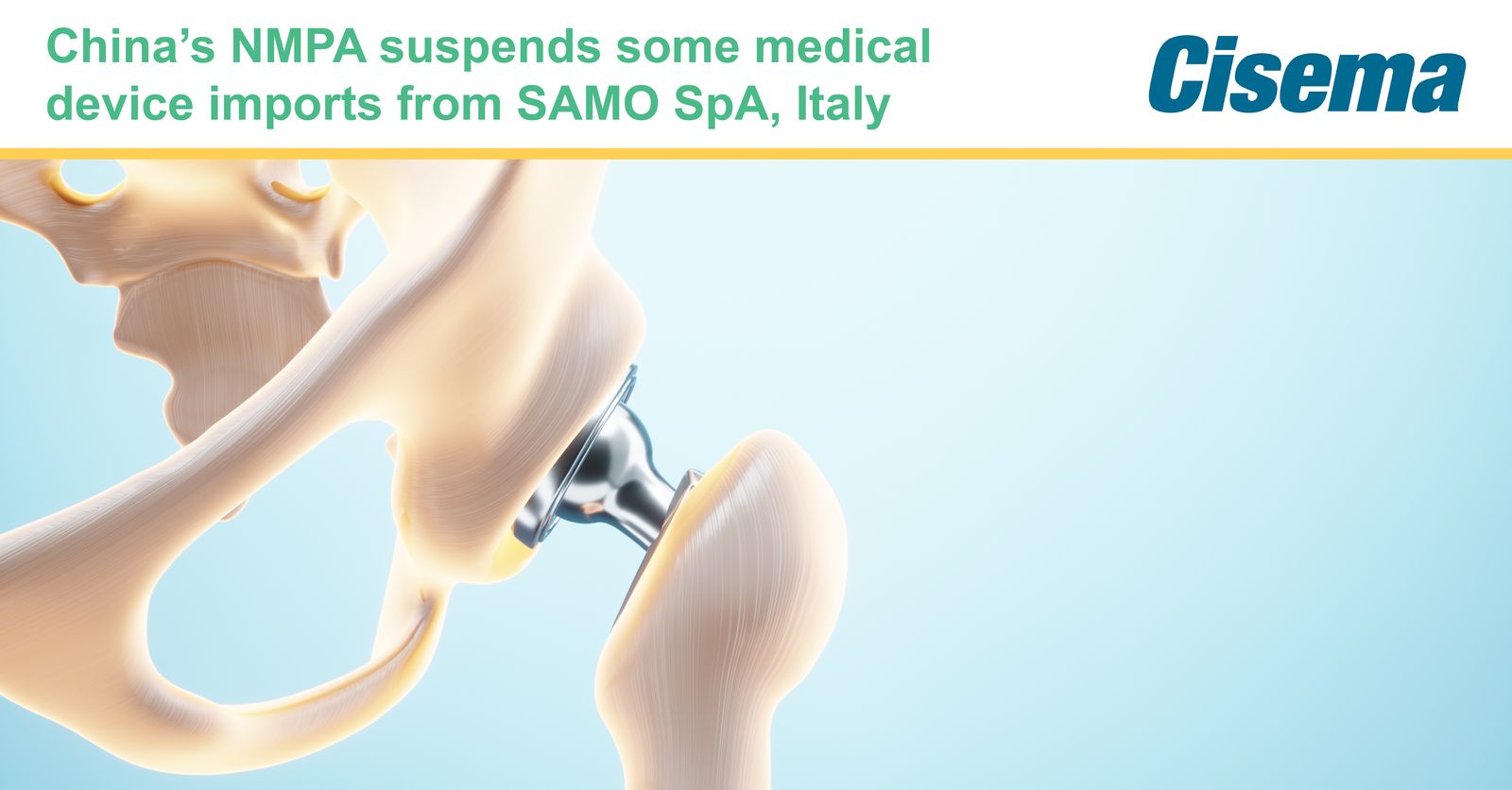
China’s NMPA has announced a suspension of import, sale and use of two medical devices manufactured by SAMO S.p.A. because they failed to comply with regulatory requirements.

China’s NMPA has issued a pilot program to optimize innovative new drug clinical trial review and approvals. in Beijing and Shanghai have been approved as the first pilot regions.

China electric bicycle charger and Lithium-ion battery CCC rules were released by the National Certification and Accreditation Administration on July 29, 2024.

MDACS listing is more crucial now for Hong Kong public procurement, especially for MedTech companies looking into entering the GBA market

China’s CMDE has issued a Draft for Comments on its latest update to the Clinical Trial Exemption List. The deadline is August 30, 2024.

China’s NMPA has issued a draft for comments on an updated Good Manufacturing Practice – GMP for Excipients and Packaging. The deadline to submit feedback is 20 August 2024.

China Medical Device Registration for June 2024 were on trend with 2023 overall approvals. Here are some charts to summarize this.

The NIFDC has released new guidelines for China Challenge Tests Cosmetics for manufacturers selling products to China.

The NIFDC has released new guidelines for cosmetic packaging compatibility testing. Other guidelines were released together on July 8, 2024.

China’s NIFDC has issued new guidelines for Cosmetic Stability Testing for cosmetic manufacturers selling products to China.

China’s Institute for Veterinary Drug Control has released 16 Veterinary Disinfectant Test Report Templates.

China veterinary anesthetic drugs and psychotropic drugs regulations have been published by the Ministry of Agriculture on July 11, 2024.

China Electric Bicycle Battery certification rules CCAP-GZ-464204:2024 have been revised and replace the previous version CCAP-GZ-464204:2023.

CCAP certification of vehicle batteries has revised rules and requirements. These were published by the CCAP China on April 30, 2024.

Post-market changes to Active Pharmaceutical Ingredients in China now have clearer guidance thanks to a recently published Q&A from CDE.

China’s NMPA has issued an update to Radio Frequency Device Requirements in China on July 8, 2024 to ensure the safety.

The NMPA is requesting comments on China drug approvals of Clinically Urgent Overseas Drugs. The deadline to submit comments is July 24, 2024.

China’s NMPA has explored numerous AI use cases in Pharma. The scenarios aim to promote AI research and AI applications in drug regulation.
Get in Touch with Our Consultants
And discover how we can support you.
