Published on
Last updated on
China Clinical Trial Exemption List Update 2024 – Draft for Comments

The CMDE has proposed an update to the China Clinical Trial Exemption List. Industry stakeholders are invited to feedback on the proposals by August 30, 2024 – see below for details on how to do so. The update includes 23 newly added products and 20 modifications to existing products already on the list.
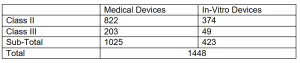
There are specific clinical evaluation exemption lists, which the NMPA publishes and updates occasionally. If a product appears on the list, it is clear that there is no need to undergo a clinical trial in China for NMPA registration and a Clinical Evaluation Report will be sufficient. The latest batch of exempted products was released in July 2023. Currently, on the China clinical trial exemption list, there are:
Products proposed for inclusion in the clinical trial exemption list
Classification Category Classification Code Product nameII02-12-00Personalized 3D printed surgical modelsIII03-13-06High-pressure balloon dilation catheter with infusion port III03-13-28Atrial septal puncture sheath III04-12-00Bone marrow infusion needle and drill II05-04 Radiation therapy equipment III05-04-00 Radioactive seeds are implanted using a puncture needle II06-14-01 Spinal endoscopy II07-03-03 Ambulatory blood pressure recorder III07-04-00Neurally monitored endotracheal intubation II07-08-01 Telemetry monitoring system II07-08-02 Remote monitoring system II07-09-07 Body composition analyzer II08-01High-flow respiratory humidification therapy device II08-05-12 Defibrillation electrodes II08-06-12 Fogging mask II09-04-02 Pre-habilitation trainer II09-04-05 Pneumatic ballistic extracorporeal pressure wave therapy deviceII17-01-05 Oral digital impression machine II17-06-03Temporary crowns, bridges II17-07-07 Custom-made appliances II22-08-01 Radioimmunoassay γ counter II22-09-06 Instruments for the analysis of genital secretions II22-10-01 Flow cytometry lattice instruments
Modifications to products already listed in the clinical trial exemption list
The proposed changes to products already listed in the medical device clinical trial exemption list concern various types of changes including changes to the classification code, the product name, and the product description. They do not include changes to the classification category.
Change of classification code:
- Head nails (change of code from 13-06-03 to 03-14-02). The product is made of stainless steel and should be used in conjunction with a skull fixator to fix the patient's head and neck in a specific position during surgical procedures requiring severe repair of the skull. The product is non-implantable, is only used intraoperatively, has short-term contact with the human body, and is removed immediately after surgery.
Change of product name:
- Non-invasive automatic blood pressure measurement to Electronic sphygmomanometer
- Multi-parameter monitor to Patient monitors
- Cooling blankets to Physical cooling equipment
Change of product description:
- There are 17 proposed changes to product descriptions. In most instances, the changes are very slight – please contact your NMPA Legal Agent to understand how these may affect your product certificates.
How to submit comments:
Send your comments using the feedback form which can be found in Annex 2 of the original announcement and send it to the email address huangyl@cmde.org.cn before August 30, 2024. Please indicate "2024 Device Exemption Catalogue Feedback" in the subject line.
Further information:
- To read the original announcement click here.
- For assistance on product classification or clinical trial exemption, please contact Cisema.
GET IN TOUCH
🌐 Send us your enquiry📚 Request our whitepapers📣 Sign up for our newsletter



