News

The CDE published the trial working procedures for initiating drug verification and testing with immediate effect from Jan 1, 2022, onwards.

A variety of consultation channels are offered by the CDE to enhance the support of technical guidance and services to applicants.

Measures for the quality management of cosmetics production will come into force on July 1, 2022. Production requirements shall be complied.

China electronic certificates for cosmetics registration was announced for trial implementation by the NMPA on December 30, 2021.

Overseas clinical trial data for IVD registration in China now adopts the new technical guidelines issued on December 1, 2021, by the NMPA.

8 GB standards and 19 YY/T standards have been approved whilst NMPA issued a draft list of 76 mandatory standards related to GB 9706.1-2020
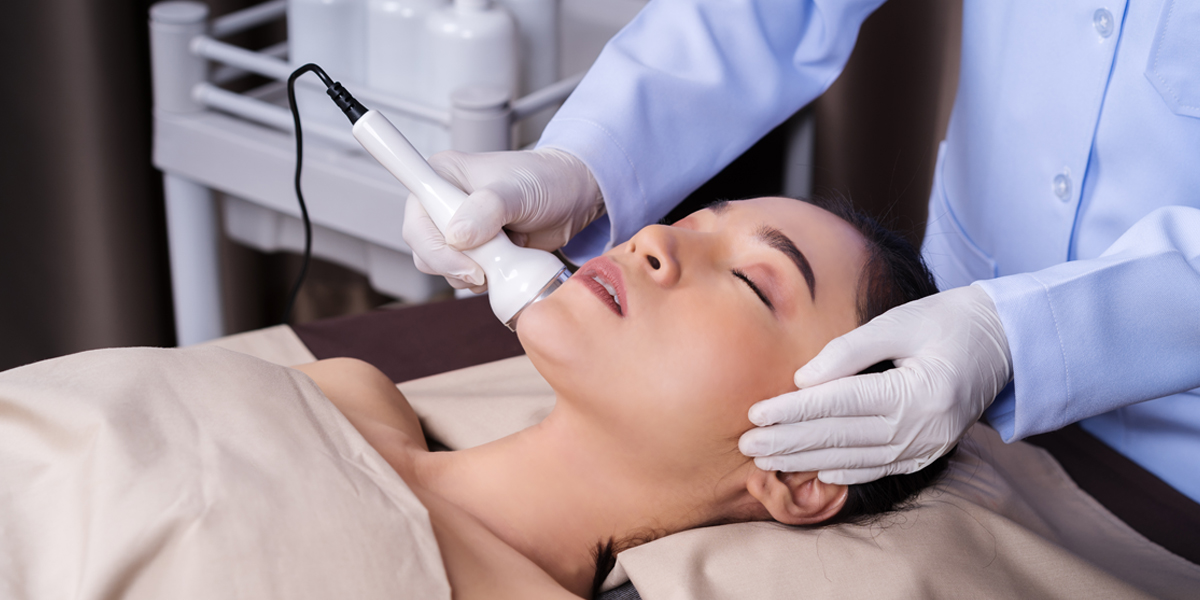
Following the 6 cases reported last month, the NMPA issued another list of violations committed by medical beauty service providers in China.

New finalized emergency approval procedures for medical devices in China with scope, requirements, application procedures, etc., clarified.
According to NMPA's new classification catalogue, non-sterile liquid & paste dressings are no longer classified as a class I in China

China healthcare market update webinar was held on Dec 15, 2021, by Cisema with the EU SME Centre and the China-Britain Business Council.

New drug clinical trial applications 2020 annual report indicated a total of 2602 clinical trial applications received by the CDE.
China innovative drug approvals require pre-NDA meetings carried out in the application to address the common pharmacology issues

Cosmetics Ingredient Submission Code is required from Jan 1, 2023, onwards to cover the safety information of cosmetic ingredients in China

New Medical Device Classification Catalogue was announced in draft on November 9, 2021, by the China NIFDC

Annual QMS self-inspection reports have new filing instructions and new report submission requirements currently in draft.

OEM quality agreement for medical devices helps to set out the the rights, obligations and responsibilities of registrants, filers and OEM...

Medical device clinical trial data submissions in China should now refer to the the NMPA technical review guidelines issued on Nov 25, 2021.
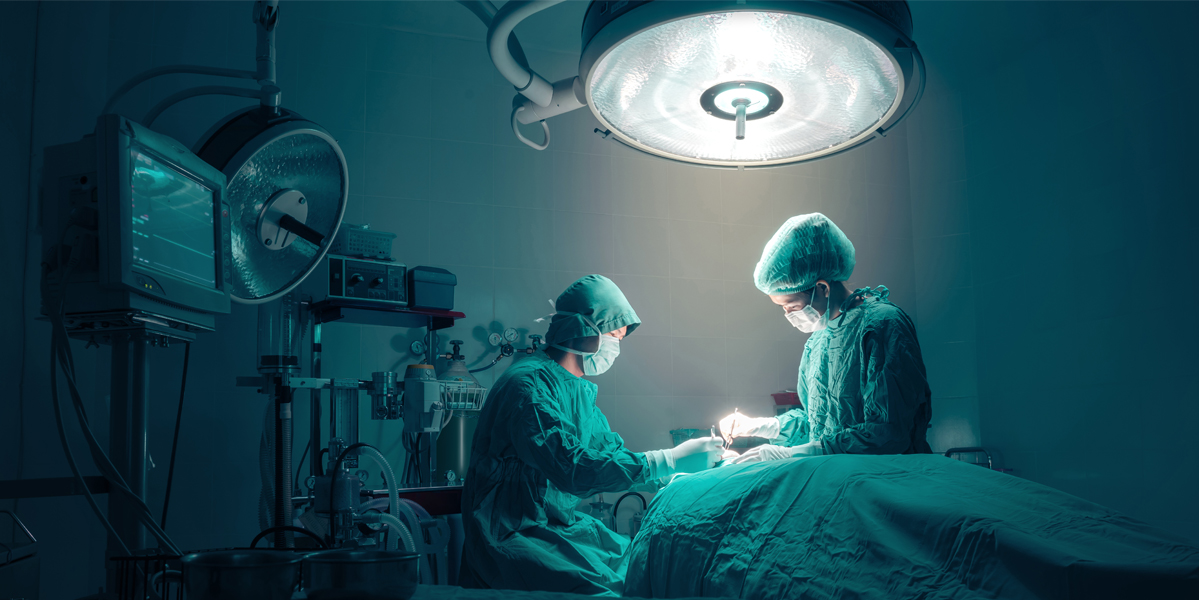
22 high-risk medical implants have been added in the draft catalogue of medical device products to be prohibited from China OEM production.

The NMPA issued a list of common violations committed by medical beauty service providers in China throughout 2020 and 2021.

Cisema presented on the topic of registration and approval of medical devices in China to 94 participants, and held a Q&A session after.
Get in Touch with Our Consultants
And discover how we can support you.
