News

Measures for Cosmetics Adverse Reaction Monitoring consists of 7 chapters covering general provisions, adverse reaction investigation, etc.
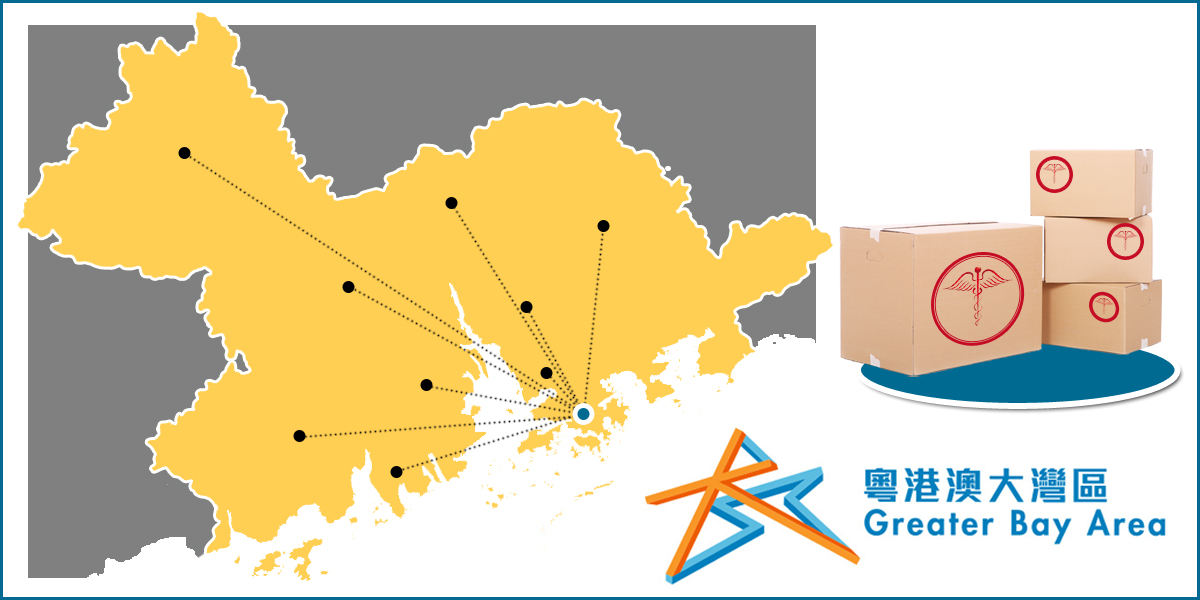
Importing drugs and medical devices from Hong Kong have been approved and amounted up to 15 products into the GBA as of March 2022.

2022 sampling inspection plan covers 60 products mainly targeting IVD reagents, anti-epidemic medical supplies, etc.

The NMPA report on China medical device standards management provided a comprehensive overview of the past year’s developments.

China issued digital health regulatory guidelines for registration of medical device software including AI, and for cyber security.

China general cosmetics urgent deadline is 31 March every year as part of CSAR's framework to file an annual report to the NMPA

Cisema Webinar on Cosmetics Registration explains ins and outs of filing/registering cosmetics products as the CSAR takes over the old CHSR

Clinical trials GMP management specification first issued its draft in July 2018. The appendix draft was issued on January 18, 2022.

Acceptance and examination of chemical APIs trial guidelines have had its first draft issued in April 2020.

Medical device product technical requirements have new guideline issued by the China NMPA on February 9, 2022.
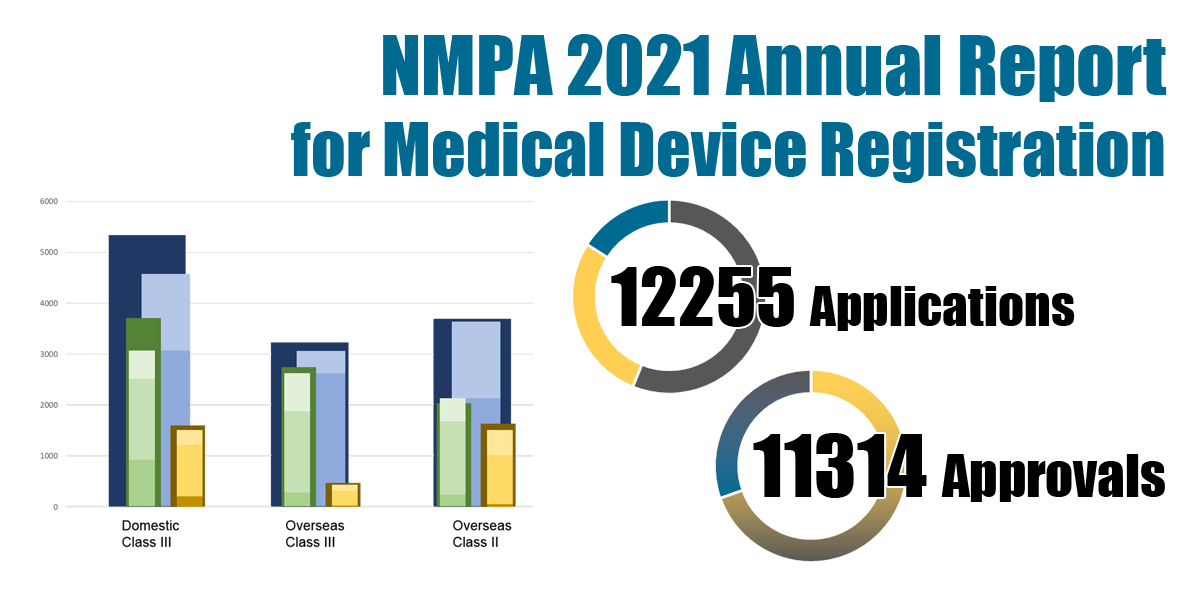
The NMPA reported a total number of 12,255 applications and 11,314 approvals for medical device and IVD registrations in 2021.
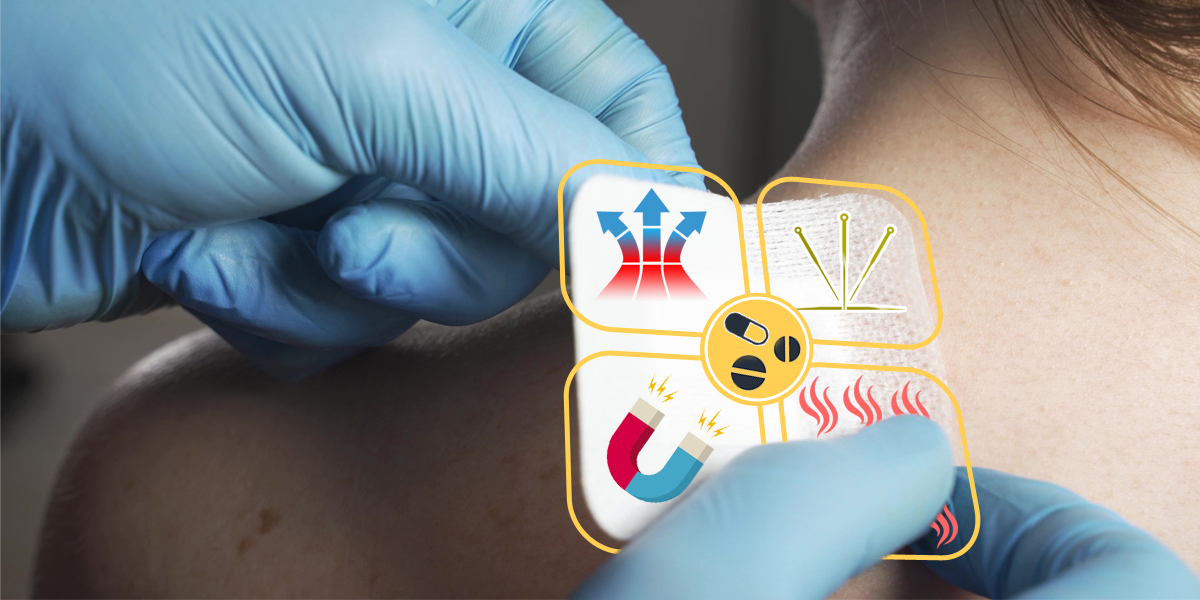
In order to restrain the illegal use of drug ingredients in medical dressing patches, the China NMPA approved the testing methods propose....

China Medical Device Standards have been recorded with a total of 1,849 as of December 2021, whilst 181 standards were newly issued in 2021.
The CNAS issued the CNAS-EL-21: 2022 to provide guidance for applying for the accreditation for group enterprise internal laboratories.

The NMPA issued the technical review guideline for medical device-based combination products, and on drug release studies required...

The NMPA published the 14th Five-Year Plan for National Drug Safety and Promotion of High Quality Development setting goals to (i) achieve...

Cisema celebrates 20th anniversary in 2022. We strive to join German efficiency and reliability in service to our customers.

Cisema webinar on drug registration pathways will provide an overview of the regulatory environment for medicines in China.

China Water Efficiency Label will be required for a 3rd batch of products in China according to the NDRC that comes into force in July 2022

The SAMR issued a draft list of Health Functions Allowed for Non-Nutrient Supplements Health Food Claims and supporting documents
Get in Touch with Our Consultants
And discover how we can support you.
