News

China YY Standards 2022 Implementation and Amendment Plan covers 59 new YY/T standards and 57 existing industry standards to be amended.

"China GMP Medical Device 2022 Edition for sterile and implantable devices" by Beijing MPA replaces the 2016 edition with immediate effect.

Webinar "China Cosmetics Ingredient Code Submission" will be held on June 15, 2022, for cosmetics manufacturers and ingredients suppliers.
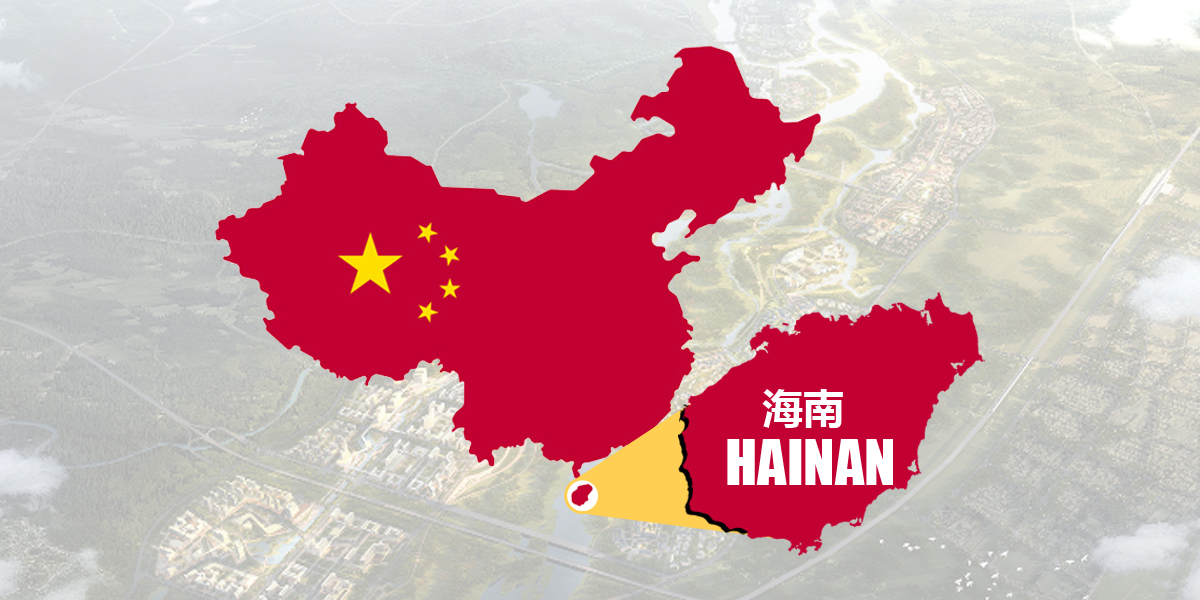
The Communication SOP for Medical Device Clinical RWD Applications (Trial) was jointly issued by the CMDE and the Hainan MPA in April 2022.

China clinical application management is an important part of the modern hospital management system to standardize medical technologies.

Cosmoprof Worldwide Bologna 2022 - Schedule an appointment to learn about registration of cosmetics and cosmetic ingredients in China

Measures for the Supervision and Administration of Drug Production was issued by the SAMR and came into effect on July 1, 2020.

Measures for the Administration of Drug Registration took effect on July 1, 2020, to reform the drug review and approval system.

The Guideline for Pharmacovigilance Inspection urge MAHs to implement the main responsibility of pharmacovigilance.

China Drug Annual Reports Administrative Regulations was issued by the National Medical Products Administration on April 12, 2022.

Technical Guideline for Children's Cosmetics sets to standardize and guide the cosmetic product registration/filing to ensure children safety

FAQ for General Cosmetics Filing is a publication first issued by the Beijing Municipal Medical Products Administration in November 2021.

Cosmetics Safety and Technical Standards have been revised by the China National Institutes for Drug Control and currently open for comments.
OEM prohibited medical devices means original equipment manufacturers (OEM) are banned from production in China for certain medical devices.
Medical device classification catalogue changes to non-sterile liquid/paste dressings, neurological/cardiovascular surgical instruments
Supervision of medical device distribution in China will be strengthened when the new regulatory measures take effect on May 1, 2022.

Supervision of medical device manufacturing in China will be strengthened when the new regulatory measures comes into effect on May 1, 2022.

The China Clinical Trials GCP (Good Clinical Practice) 2022 amended version will come into effect on May 1, 2022.

China GB 16897 for brake hoses and tubes of vehicles (automobile, motorcycle and trailer) was proposed for implementation on July 1, 2023.

China NHC announced the approval of 2 new food ingredients, 11 new food additives, and 19 new food-related product varieties.
Get in Touch with Our Consultants
And discover how we can support you.
