News

The CMDE published several interesting case studies to address some of the detailed requirements for IVD and SiMD registration in China

The NMPA is revising the Class I Medical Device Classification Catalogue to cover 600 new product examples.

China clarifies rules for registration and classification of drug-device combinations and read more about the NMPA

On January 1, 2022, the China Measures for the Supervision and Administration of Production and Operation of Cosmetics will be implemented

Due to stricter risk managements imposed by the EU MDR, there could be a high approval rate for EU medical devices registering in China.

The NMPA implemented the “Notice on Matters related to the Registration of Drug-Device Combination Products” (No.52-2021).

NMPA revised the draft “Required Working Measures on Self-Testing for the Registration of Medical Devices” to support Order 739.

The draft amendment covers the guiding principles for the shelf-life of passive implantable medical device for registration and filing.
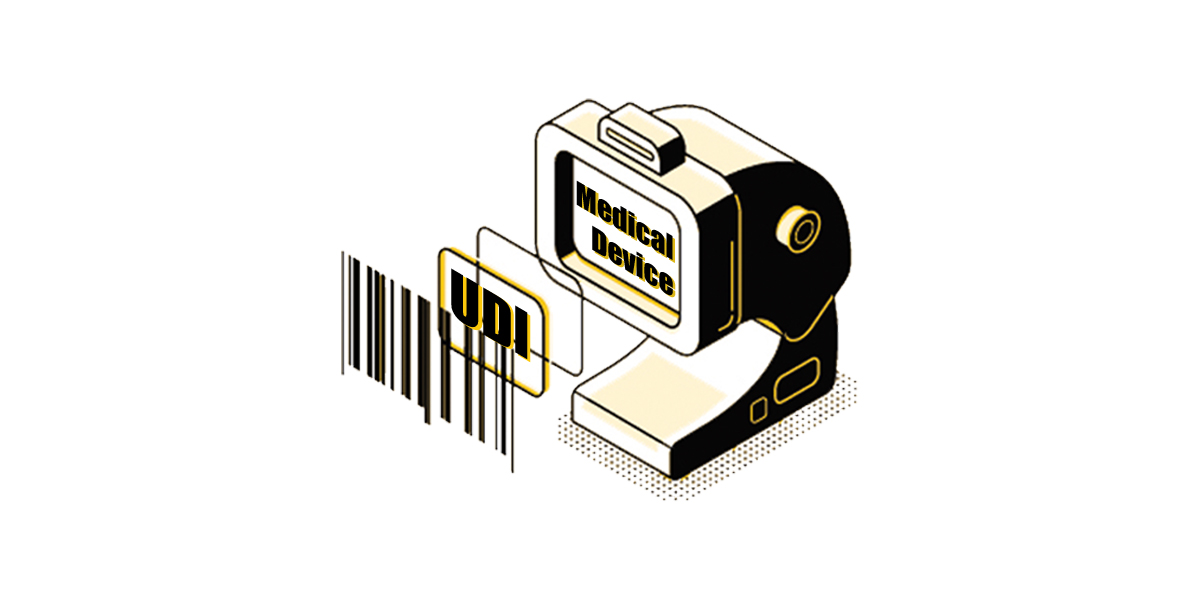
NMPA published a regulatory draft to require all class III medical devices and IVDs manufactured after March 1, 2022 to have UDI.

NIFDC announced 5 drug-based combination products and 5 medical device-based combination products among a total of 22 application results.

On July 8, 2021, the China National Medical Products Administration announced the classification guidelines for AI medical devices and SaMDs.
Popular beauty devices are classified as medical devices according to the classification catalogue in China
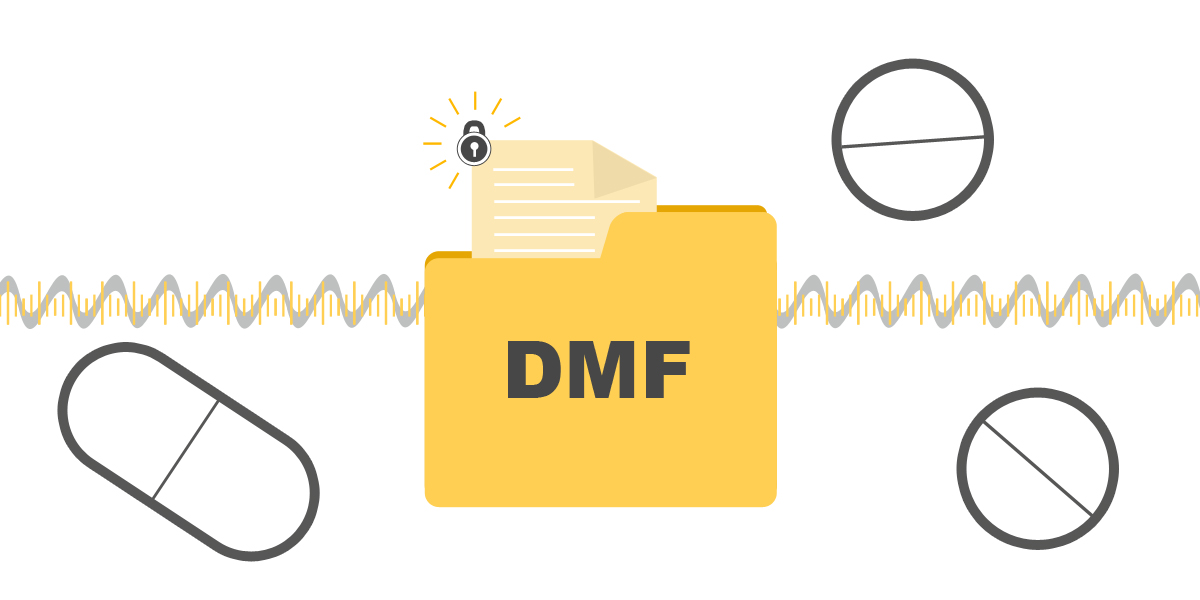
Drug Master Filing permits APIs, excipients and packaging materials manufacturers protect intellectual property and market approval.
Biocompatibility testing is the one test that can be done outside of China provided it complies with the GB/T 16886 and ISO 10993 standards.

The NMPA regulatory draft imposes stricter controls and raises the safety standards on Children's cosmetics under the age of 12.

Grace period will be around 11 months for new applicants, and around 23 months for applicants with approvals before May 1, 2022.
The NMPA (National Medical Products Administration) issued the Catalogue of Technical Guidelines for Reviewing Medical Device Registrations.

The catalogue covers 1,791 medical industry standards that are currently in effect. 77 industry standards will be revised this year.

List of institutions qualified for conducting clinical trials and also completed the filing application to be listed in the NMPA database.

New revised law on the Supervision and Administration of Medical Devices (Order 739) issued by the State Council came into force in June 2021
Get in Touch with Our Consultants
And discover how we can support you.
