News
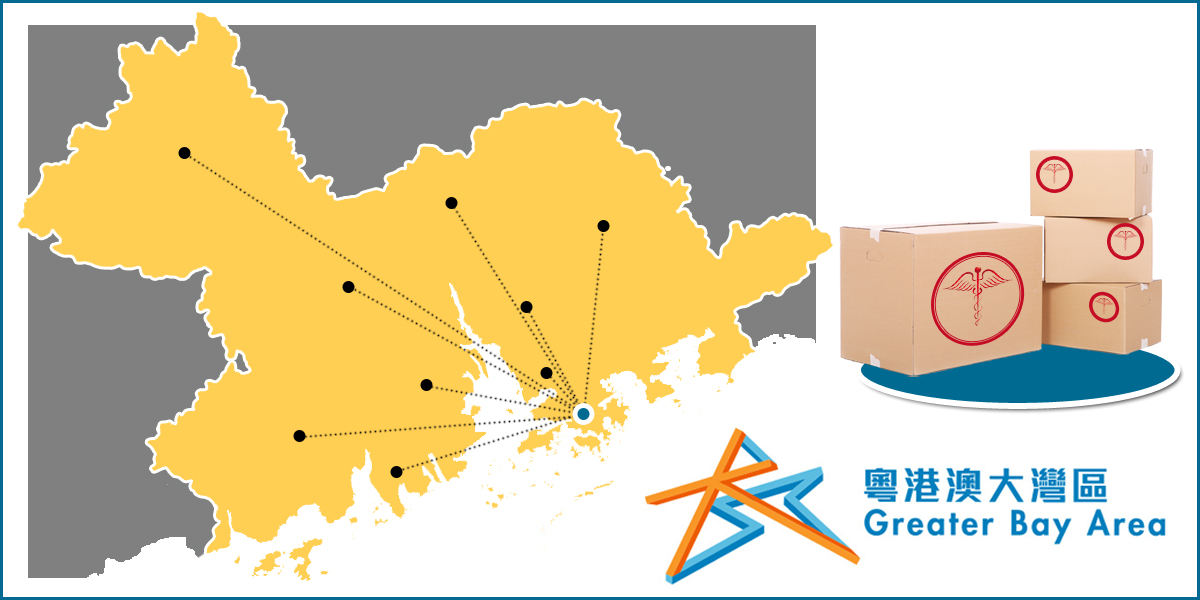
The Guangdong MPA announced the first approved medical device arriving the University of Hong Kong – Shenzhen Hospital via the Greater Bay Area initiative.

The SAMR published new technical requirements to expand the scope of health food in dosage forms and excipients for filing, with effect from June 1 onwards.

The NMPA announced 2 new Provisions to support the implementation of the CSAR, and the exemption of animal testing for general cosmetics.

On March 2, 2021, the NMPA announced 7 major updates on the Safety and Technical Standards for Cosmetics (2015 version) with effect from May 1 onwards.

22 technical guidelines (draft) were issued in March 2021, and technical guidelines for products monitoring non-invasive blood / glucose will be drafted
The NHC announced the implementation of The Administrative Measures for the Clinical Use of Medical Devices coming into force from March 1, 2021 onwards.

The Guangdong MPA (Medical Products Administration) announced details about the pilot plan and the responsibilities of different departments involved.

The NMPA announced the implementation of a mNMPA Introduces the Medical Device Master Filing System covering both medical devices and IVDs.

First remote inspection successfully carried out in January 2021 for SELO China manufacturer license, and many more to come.

The NMPA announced a total of 28 updates on the classification catalogue for medical devices including 15 classification changes and 13 content updates.

AdvaMed advocates globally for the highest ethical standards and patient access to safe, effective and innovative medical technologies that improve lives.

On December 4, 2020, the CNCA announced the temporary guidelines for sampling testing on five groups of organic food products in China.

2 amendment drafts concerning the prohibited list of cosmetic ingredients & the IECIC (Inventory of Existing Cosmetic Ingredients in China) were released.

6 technical guidelines & 4 GB / YY draft standards were announced; 2 products registered & 7 applications accepted by the fast-track approval pathway.

On February 5, the NMPA issued a report covering a breakdown of the total existing and new standards for medical devices in 2020.

On January 19, the NMPA announced the new clinical trial exemption list for medical devices and in-vitro diagnostics reagents.
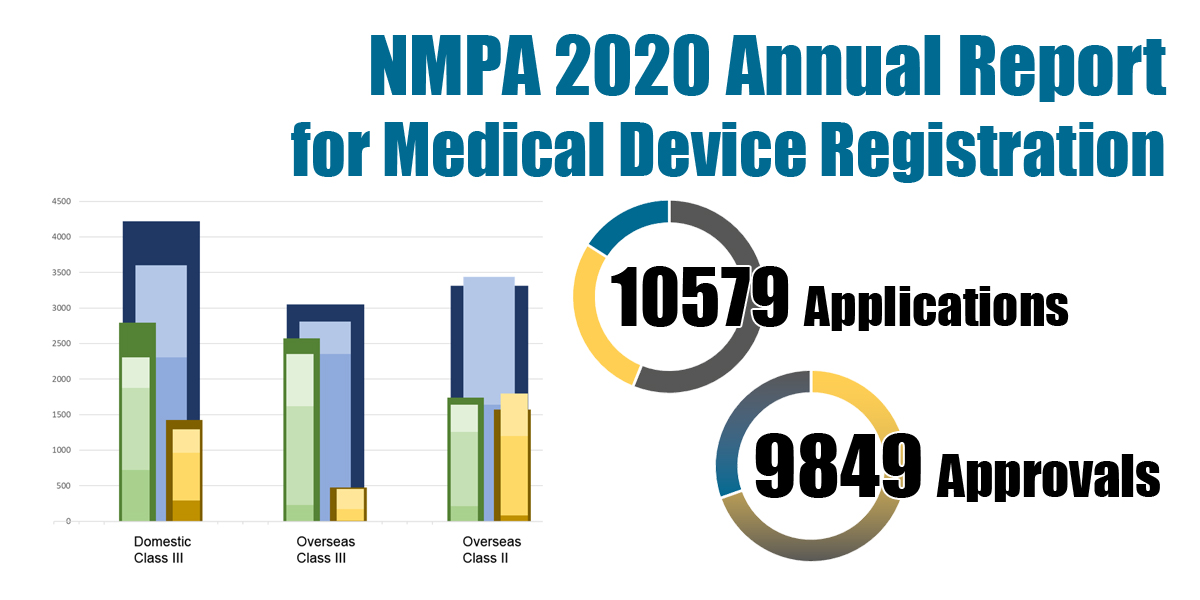
A total number of 10579 applications and 9849 approvals for medical device registrations in China were reported in 2020 along with a detailed breakdown.

The SAMR addressed the basic requirements for registration and filing management of cosmetics and new cosmetic ingredients in China to begin on May 1, 2021.

LifeSciences BC is a industry association that supports and represents the life sciences community of British Columbia in Canada.

The National Health Commission announced new food ingredients and additives are permitted to be used in food-contact articles.
Get in Touch with Our Consultants
And discover how we can support you.
