Medical Devices
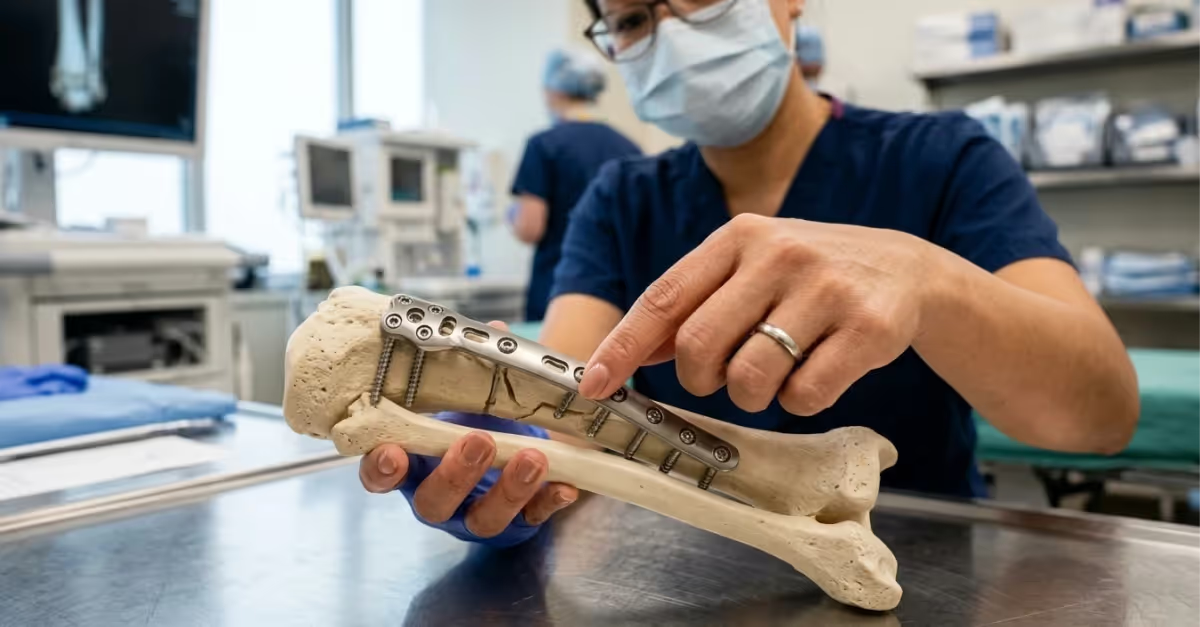
The 26 new medical device standards cover orthopedic implants, IVDs, active devices, and software risk management.

China’s NMPA mandates full UDI coverage for all medical devices by 2029, linking compliance to registration, reimbursement, and market access.

China’s NMPA issues new guidance on clinical trial protocol pre-review for innovative devices, impacting overseas fast-track approvals.

China NMPA’s 2025 report highlights growth in medical device approvals, including advanced imported devices.

China’s NMPA releases 2025 annual report on medical device standardization, highlighting growth, innovation, and international alignment.

The draft 2026 catalogue defines which Class III medical devices would require clinical trial approval before trial initiation.

Malaysia’s MDA withdraws personal use medical device importation Guidance Document, directing stakeholders to updated exemption requirements.

Vietnam’s MoH recently published Circular drops several healthcare advertising regulations and streamlines requirements for many categories.

Malaysia’s MDA and Thailand’s FDA launch a medical device reliance pilot to accelerate market access for Class B–D devices.

Malaysia clarifies that medical device tenders count as market placement, triggering licensing and compliance requirements.

Malaysia’s MDA will regulate aesthetic and cosmetic medical devices under the Medical Device (Designated Medical Devices) Order 2026.

Circular No. 1/2026 clarifies regulatory requirements for medical device procurement tenders, such as licensing and GDPMD compliance.
Get in Touch with our Certifications team
And discover how we can support you in getting your products certified for China.
