Published on
Last updated on
Chinese Medical Device Regulatory Data: 2024 Summary and Analysis

On January 10, 2025, the National Medical Products Administration (NMPA) released a detailed report on medical device registrations and licensing activity across China. This publication, which reflects data up to December 31, 2024, is part of an ongoing effort to improve regulatory transparency and foster public access to information.
The NMPA's initiative supports better public oversight and shared governance while highlighting the extensive scope of China's medical device industry. Regular monthly updates will be available on the NMPA’s official website via its data query page.
Overview of Chinese Medical Device Regulatory Data
The report outlines a total of 126,139 registered medical device products and 163,489 filing certificates issued across the country. Additionally, authorities granted 19,714 production licenses to manufacturers and 23,984 operational licenses to businesses in distribution.
E-commerce activity has surged, with 483,479 network sales licenses issued alongside 1,458,567 filings for third-party online platforms. These figures reflect the growing scale of the digital healthcare economy and the importance of regulatory oversight in this area.
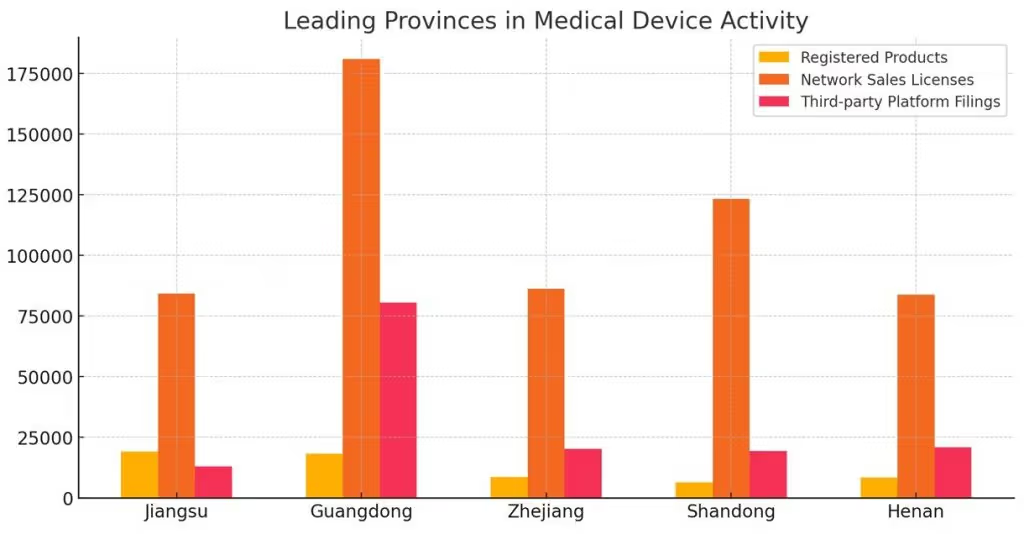
Leading Provinces in Medical Device Activity
Several provinces continue to lead in medical device activity. Jiangsu reported 19,071 registered products and accounted for 84,124 network sales licenses. Similarly, Guangdong emerged as a leader in both production licensing, with 3,066 licenses, and third-party platform filings, which reached 180,846 records.
Other regions, such as Zhejiang, also showed substantial activity, particularly in operational licensing and e-commerce infrastructure development. These provinces are key hubs for medical device innovation and manufacturing in China.
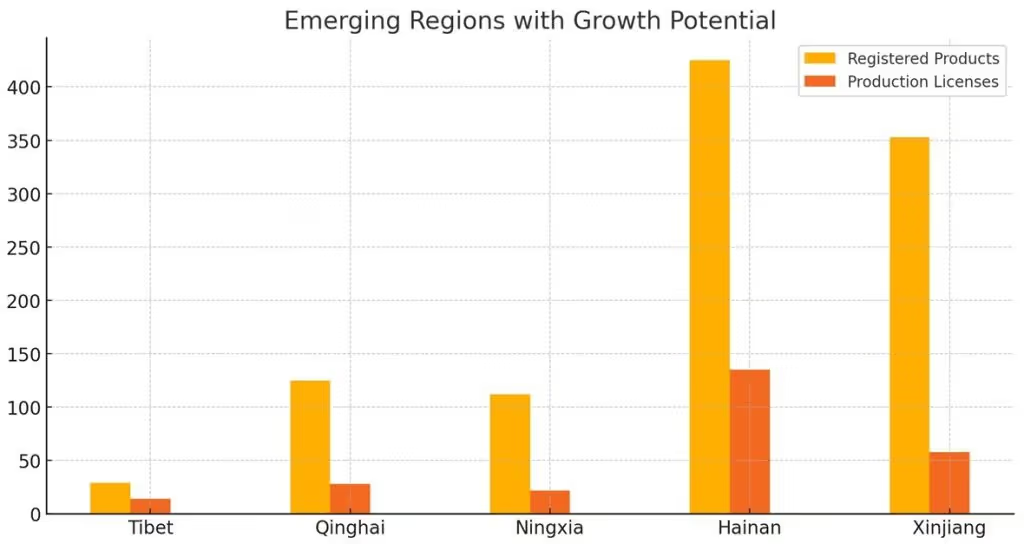
Emerging Regions with Growth Potential
Smaller provinces such as Tibet, Qinghai, and Ningxia showed modest but notable figures in the dataset. Tibet recorded 29 product registrations and 14 production licenses, while Qinghai filed 125 product registrations and issued 28 production licenses. Ningxia added 112 product registrations and granted 22 production licenses.
While these numbers are lower compared to larger provinces, they indicate potential for future growth, particularly as healthcare infrastructure develops. Hainan, though a small province, recorded 11,588 network sales licenses, highlighting its growing role in the digital distribution of medical devices.
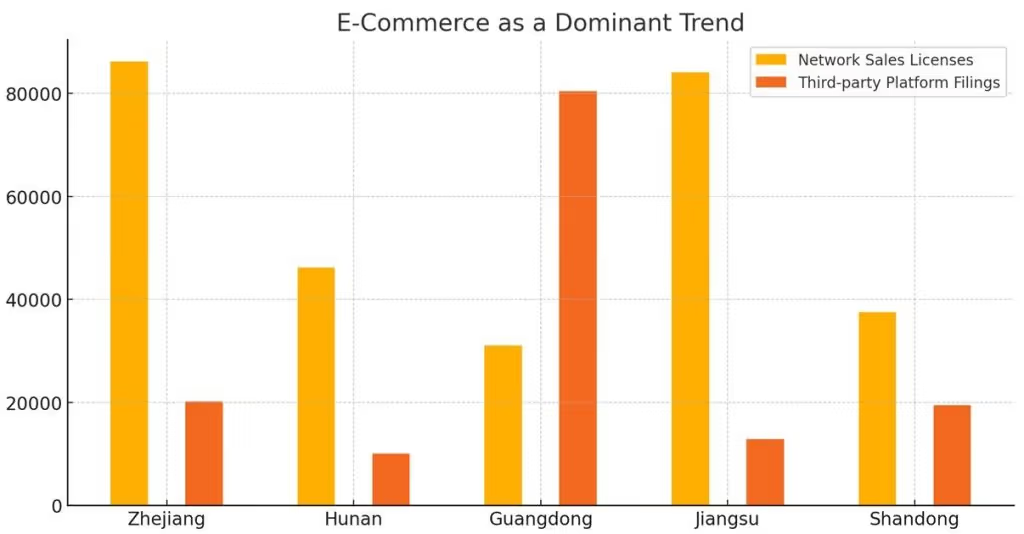
E-Commerce as a Dominant Trend
E-commerce continues to reshape the medical device industry in China. Zhejiang issued 86,205 network sales licenses and reported 20,169 third-party platform filings, highlighting its strong digital commerce ecosystem. Meanwhile, Hunan contributed significantly with 46,190 network sales licenses and 10,016 filings for third-party platforms.
These figures illustrate the rapid integration of digital channels into the medical device supply chain. The reliance on e-commerce and third-party platforms emphasizes the need for consistent regulatory oversight to ensure compliance in this evolving landscape.
Commitment to Transparency and Collaboration
The report emphasizes the NMPA's commitment to administrative transparency. Businesses and stakeholders are encouraged to access the database for detailed information and report any inaccuracies or discrepancies through the correction mechanisms outlined on the website.
This approach not only strengthens confidence in the regulatory framework but also encourages collaborative efforts to maintain data accuracy and compliance.
Insights for International Stakeholders
This Chinese medical device regulatory data provides valuable insights for international compliance managers and regulatory professionals exploring the Chinese market. Established provinces like Guangdong and Jiangsu remain vital for manufacturing and innovation, while regions such as Zhejiang and Hunan drive the expansion of e-commerce.
The figures suggest opportunities for partnerships in provinces that exhibit high regulatory activity and a strong digital infrastructure. Emerging regions such as Tibet, Qinghai, and Ningxia present longer-term potential as local demand for healthcare infrastructure and products increases.
Global Alignment and Future Outlook
The NMPA’s commitment to publishing Chinese medical device regulatory data regularly signals an effort to align its practices with international standards of regulatory transparency. This approach makes China a more accessible and predictable market for medical device companies. For more specific inquiries or data verification, stakeholders are encouraged to utilize the NMPA's data query portal, which serves as a central hub for accessing detailed and updated regulatory information.
Further Information
To read the NMPA's detailed report regarding medical device license filing in each Chinese province, please click here.
If you are interested in innovative medical devices registration, please contact Cisema for more information.
GET IN TOUCH
🌐 Send us your enquiry📚 Request our whitepapers📣 Sign up for our newsletter



