Published on
Last updated on
2024 National Cosmetic Sampling Inspection Annual Report Interpretation

The 2024 National Cosmetic Sampling Inspection Annual Report released by the National Medical Products Administration (NMPA) highlights the current state of product quality and regulatory compliance in China's cosmetics market. This year’s report not only offers a detailed statistical breakdown of inspections and non-compliance cases — including imported products — but also signals key regulatory priorities that international cosmetics companies can use to refine compliance strategies and anticipate future trends.
Background of the Annual Report and Changes in the Regulatory Environment
Before delving into the details of the annual report, it is helpful to understand its background and the changes in China's cosmetics regulatory environment. In recent years and especially since the implentation of the Cosmetics Supervision and Administration Regulation (CSAR) in 2021, China has continuoued to strengthen its cosmetics regulation to ensure consumer health and safety and to promote high-quality industry development.
The 2024 National Cosmetics Sampling Inspection Annual Report is issued annually and compiles the sampling inspection data and results for the entire preceding year. The Report provides detailed quality information for stakeholders, including authorities, companies, industry associations, and consumers.
Core Content and Key Findings of the Annual Report
The annual report covers a wide range of key information, including the scope of sampling inspections, inspection items, and the discovery of non-compliant products. In terms of the scope of sampling inspections, it covers cosmetics of different categories and from different channels, ensuring the breadth and representativeness of the inspection results. The inspection items focus on ingredients closely related to consumer health, such as heavy metals, microorganisms, and banned substances, reflecting the regulatory focus and direction.
The annual report provides a detailed list of non-compliant products, reasons for non-compliance, and information on the manufacturers involved. These data serve as a reminder formanufacturers about appropriate product quality control. By analyzing non-compliant products, manufacturers can understand common quality issues and risk points, thereby taking precautions in their own production and quality management systems. For example in 2024, a total of 21,362 batches of products were sampled throughout China, of which 20,889 batches met the regulations, or 97.79%.
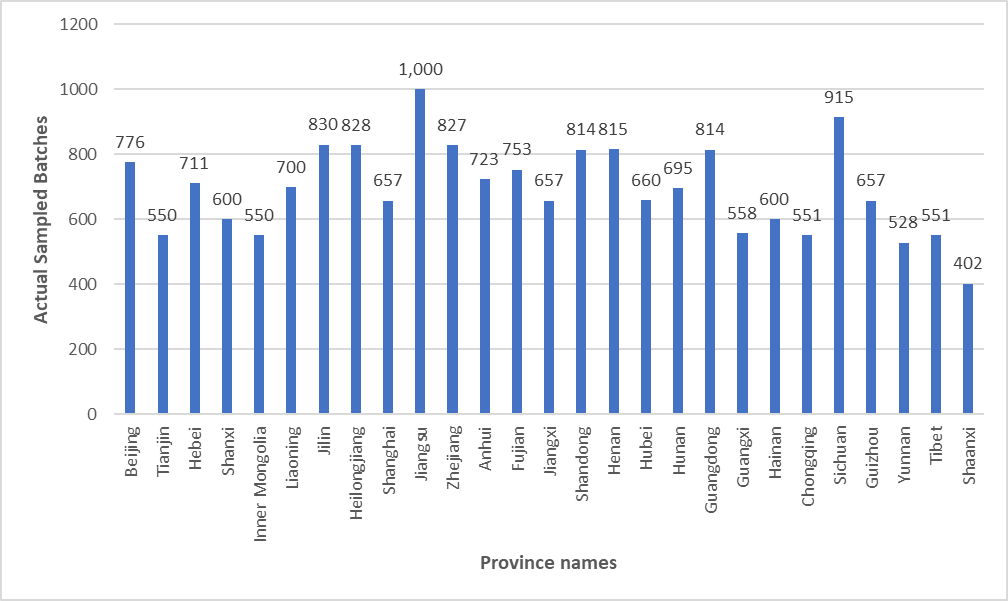
Figure 1 Sampling Distribution of Cosmetics Across Provinces (Regions, Municipalities)
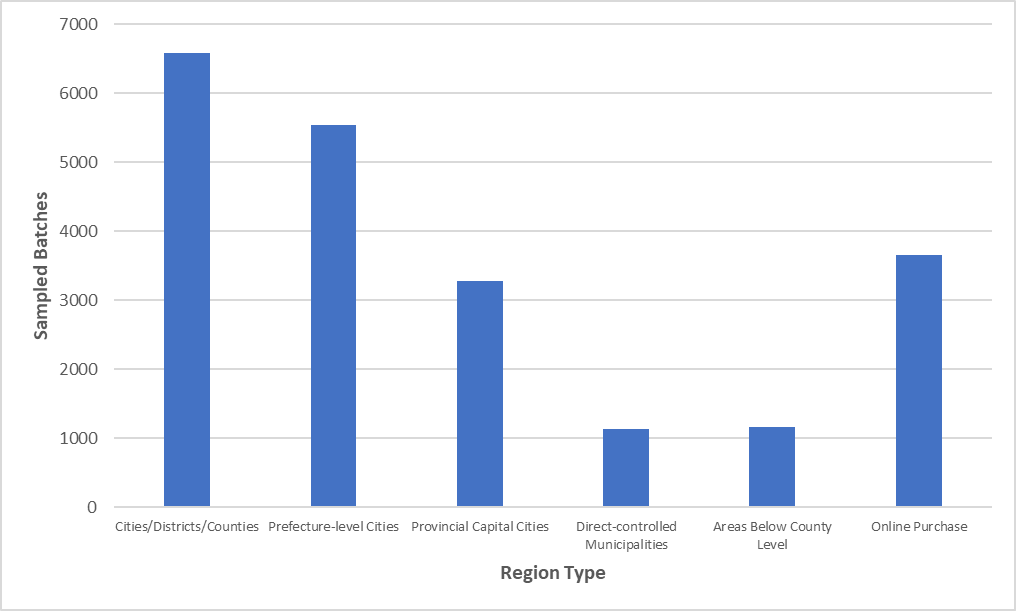
Figure 2 Sampling Quantity Distribution by Region Type
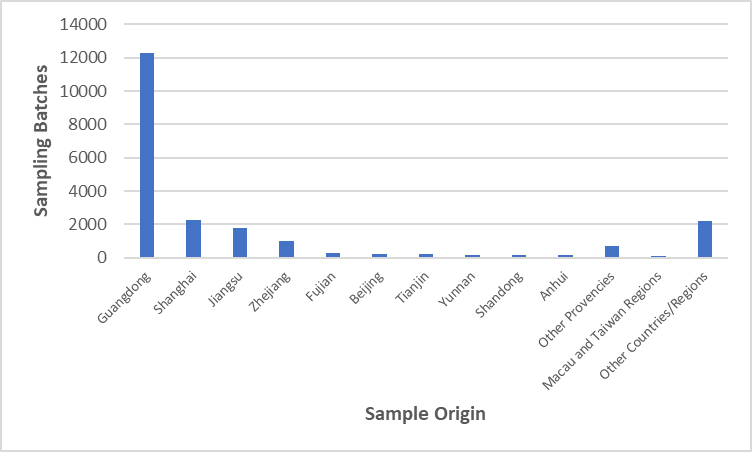
Figure 3 Distribution of Sample Origins (by Location of Cosmetics Manufacturing Enterprises)
Note: The remaining provinces in China include 18 provinces (regions, municipalities) such as Hubei, Shaanxi, and Hunan.
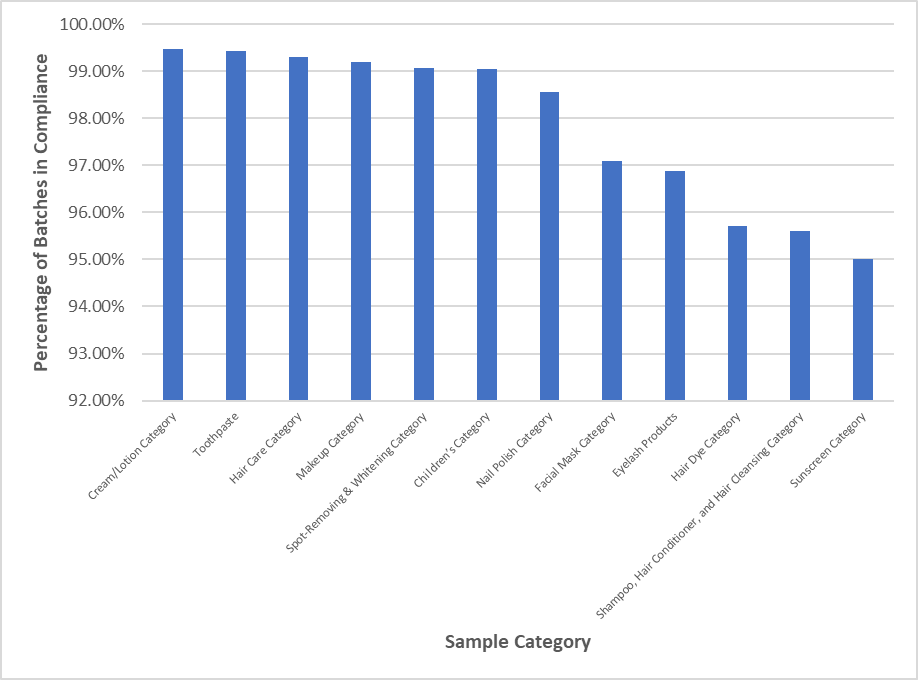
Figure 4 Proportion of Batches in Compliance for 12 Categories of Cosmetics
Sunscreen products faced the lowest percentage compliance results from inspections. Readers may detect similar trends in other markets such as recent reports by Australia's ABC of sunscreen testing failures by a consumer choice group.
Insights for International Companies
For international companies, this annual report contains some helpful insights. Firstly, it reveals the current quality status and regulatory trends of China's cosmetics market, helping international companies better understand the rules and requirements of the target market. Secondly, in terms of imported cosmetics inspections, the annual report provides relevant data and case studies, enabling international companies to clearly understand the quality challenges and compliance risks their products (and their competitors’) face in the Chinese market.
Moreover, international companies should also pay attention to the quality concerns and expectations of Chinese consumers reflected in the annual report. This can help international companies adjust their product strategies and market positioning to better meet the needs of Chinese consumers. At the same time, the annual report also serves as an important reference for international companies competing with domestic Chinese companies. By comparing the quality performance of domestic and international cosmetics, international companies can identify their strengths and weaknesses, thereby optimizing product competitiveness.
Compared to previous regulatory measures, the release of the annual report places greater emphasis on the collection, analysis, and transparency of data. This not only enhances the scientific and effective nature of regulation but also provides more information support for companies and consumers. The new issues and trends reflected in the annual report also provide reference for the revision and improvement of future regulations, helping to continuously optimize China's cosmetics regulatory system.
Further Information
To read the original announcement about the 2024 National Cosmetics Sampling Inspection Annual Report released by China NMPA, please click here.
If you are a cosmetics manufacturer with questions about how the 2024 inspection results may impact your compliance strategy for the China market, please contact Cisema.



