Published on
Last updated on
CDE 2024 Drug Review Report: Growth in Drug Registration and Regulatory Developments in China

In 2024, the Center for Drug Evaluation (CDE) accepted 19,563 drug registration applications, marking a 5.73% increase from the previous year as reported in its recently issued 2024 Drug Review Report. The data highlights continued momentum in China’s drug evaluation framework, with notable growth in chemical raw material applications and clinical trial submissions. This development is particularly relevant for international pharmaceutical companies seeking timely access to the Chinese market, as the regulatory environment continues to emphasize technical evaluation, accelerated approval pathways, and communication mechanisms.
This article summarises various other information from CDE’s lengthy annual report, which was released on March 18, 2025.
1. Acceptance of drug registration applications
1.1 Overview
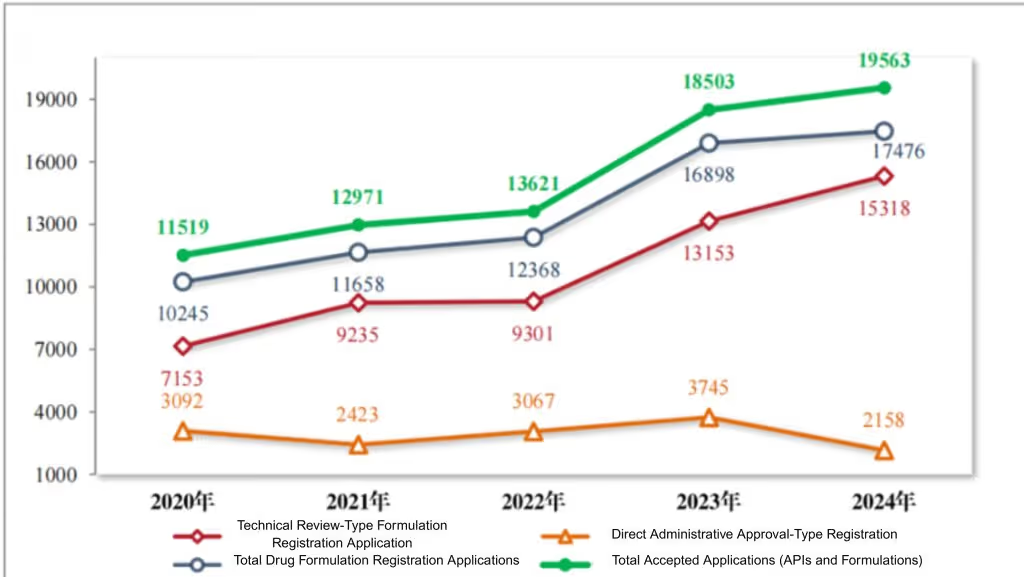
Figure 1 Acceptance of registration applications from 2020 to 2024, as reported in CDE’s 2024 Drug Review Report
1.2 Acceptance of applications for registration of drugs for technical review
Among the 15,318 technical evaluation drug registration applications accepted in 2024, there were 2,407 traditional Chinese medicine applications, 10,464 chemical drug applications, and 2,447 biological product applications. For details on the acceptance of technical evaluation registration applications for each drug type from 2020 to 2024, see Figure 2.
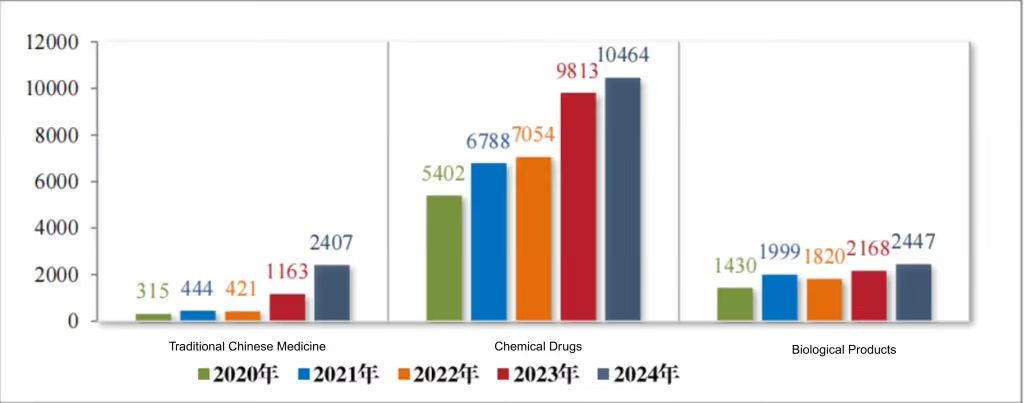
Figure 2 Acceptance of applications for registration of various drug types requiring technical review from 2020 to 2024 (cases), as reported in CDE’s 2024 Drug Review Report
From the perspective of registration application categories, 3,073 new drug clinical trial applications (IND), 247 confirmatory clinical trial applications, 549 new drug marketing authorization applications (NDA), 4,770 marketing authorization applications (ANDA) for drugs with the same name and prescription and chemical generics, 659 registration applications for generic drug quality and efficacy consistency evaluation, 5,600 supplementary applications, and 420 re-registration applications for overseas produced drugs. Compared to previous years, see Figure 3. For example, the number of IND applications for chemical drugs has increased, reflecting the continuous enthusiasm of chemical drug research and development.
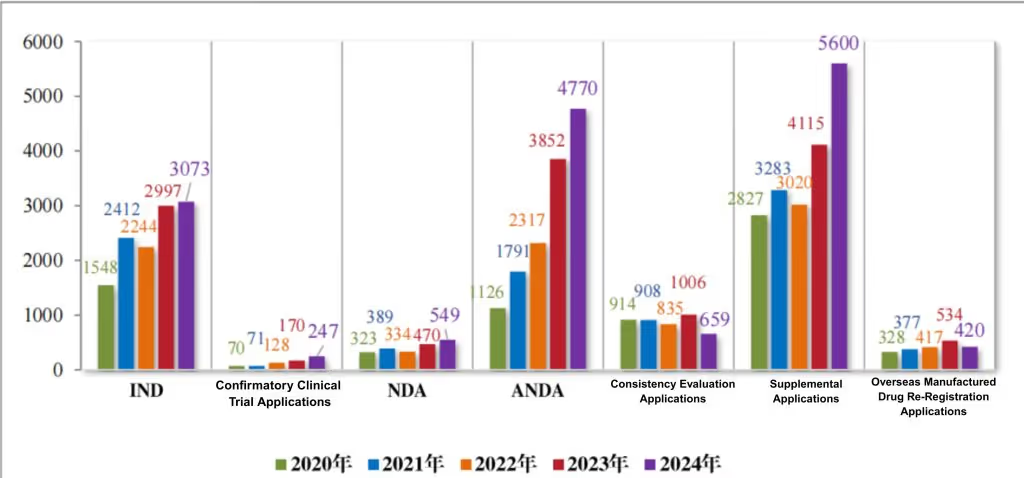
Figure 3 Acceptance of applications for registration by category subject to technical review from 2020 to 2024 (cases), as reported in CDE’s 2024 Drug Review Report
1.2.1 Acceptance of chemical drug registration applications
Of the 10,464 chemical drug applications requiring technical evaluation in 2024, there were 1,785 INDs, 247 confirmatory clinical trial applications, 255 NDAs, 4,768 ANDAs, 659 bioequivalence evaluation applications, 2,396 supplemental applications, and 354 foreign-produced drug re-registration applications. For details on the acceptance of technical evaluation applications for chemical drugs from 2020 to 2024, see Figure 4.
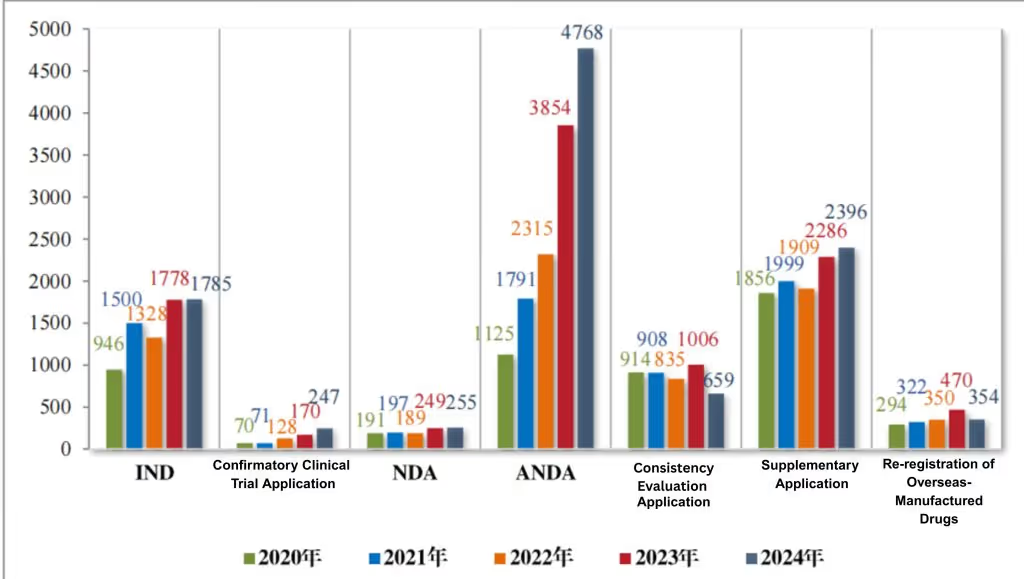
Figure 4 Number of applications for registration of various categories of chemical drugs subject to technical review from 2020 to 2024 (pieces), as reported in CDE’s 2024 Drug Review Report
Among the 1,785 INDs, 1,345 were for innovative chemical drugs (568 varieties) and 440 for improved-type chemical drugs (237 varieties). Among the 255 NDAs, 57 were for innovative chemical drugs (38 varieties), 108 for improved-type chemical drugs (46 varieties), and 90 for Class 5.1 chemical drugs (57 varieties).
1.2.2 Acceptance of applications for registration of biological products
In 2024, 2,447 biological product registration applications were accepted, including 239 for preventive biological products, 2,201 for therapeutic biological products, and 7 for in vitro diagnostics.
By application type, there were 1,188 INDs, 254 NDAs, 942 supplemental applications, and 63 foreign-produced drug re-registration applications. For details on the acceptance of technical evaluation applications for biological products from 2020 to 2024, see Figure 5.
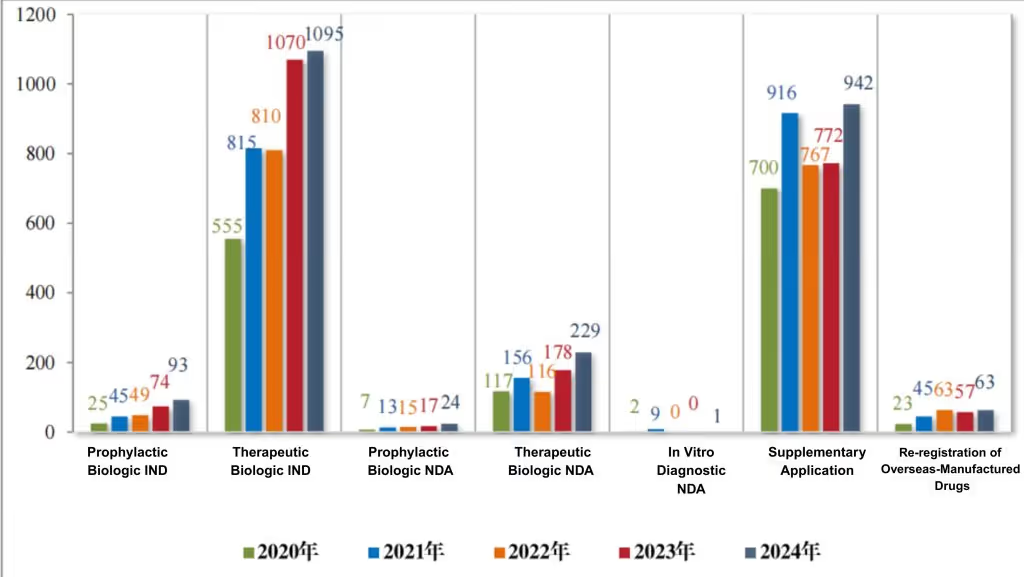
Figure 5 Acceptance of applications for registration of various categories of biological products subject to technical review from 2020 to 2024 (cases), as reported in CDE’s 2024 Drug Review Report
According to the statistics of registration classification, there were 93 INDs of biological products for prophylaxis, including 62 INDs (41 varieties) of innovative biological products for prevention, 11 INDs of improved biological products for prophylaxis (8 varieties), and 20 INDs (16 varieties) of biological products for prophylaxis that have been marketed domestically or overseas. Among the 24 NDAs for prophylactic biological products, 4 NDAs (3 varieties) for improved prophylactic biological products and 20 NDAs (14 varieties) for biological products for prophylactic use that have been marketed in China or abroad.
According to the statistics of registration classification, among the 1095 INDs of therapeutic biological products, 879 INDs (563 varieties) of innovative therapeutic biological products, 138 INDs (65 varieties) of improved therapeutic biological products, and 78 INDs (43 varieties) of therapeutic biological products marketed in China or abroad. Among the 229 NDAs for therapeutic biological products, 49 NDAs (33 varieties) were for innovative therapeutic biological products, 84 NDAs (42 varieties) for improved therapeutic biological products, and 96 NDAs (51 varieties) for therapeutic biological products that had been marketed domestically or overseas.
1.3 Acceptance of registration applications for direct administrative approval
In 2024, 2,158 registration applications were accepted for direct administrative approval. These included 1,748 supplemental applications not requiring technical evaluation and 410 temporary import applications.
2. Review and approval of drug registration applications
2.1 Overall Closure of Trials
In 2024, the CDE concluded 18,259 drug registration applications, a 16.20% increase from the previous year. This included 16,117 drug formulation applications (up 10.98%) and 2,142 chemical raw material applications (up 80.00%). Of the drug formulation applications, 13,913 were for technical evaluation (a 30.74% increase) and 2,204 were for direct administrative approval. By the end of 2024, there were 12,323 applications under review and 2,239 pending applicant responses for additional information.
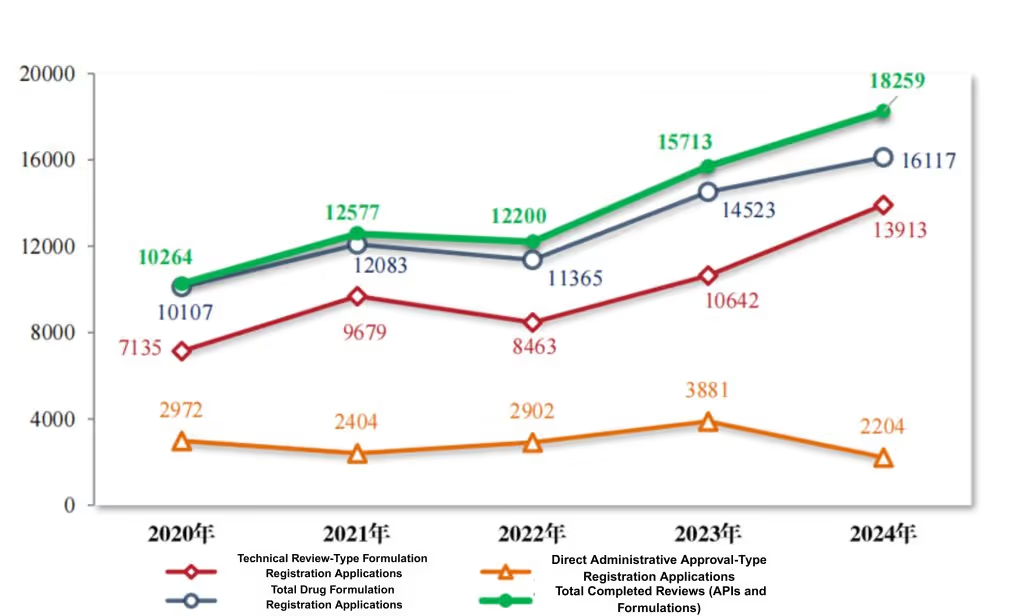
Figure 6 Number of registration applications closed from 2020 to 2024 (cases), as reported in CDE’s 2024 Drug Review Report
2.2 Completion of technical review registration applications
Of the 13,913 technical evaluation applications concluded in 2024, 1,907 were for traditional Chinese medicine, 9,759 for chemical drugs, and 2,247 for biological products. By application type, 3,011 INDs, 215 confirmatory clinical trial applications, 439 NDAs, 3,552 ANDAs, 1,135 bioequivalence evaluation applications, 5,090 supplemental applications, and 471 foreign-produced drug re-registration applications were concluded.
A total of 48 innovative drug varieties (Class 1) were approved for marketing in 2024 (see Annex 1 from the original CDE news). Of these, 17 (35.4%) were approved through the priority review and approval process, 11 (22.9%) were conditionally approved, and 13 (27.1%) were included in the breakthrough therapy program during clinical trials.
In 2024, 55 rare disease drugs were approved (excluding Class 4 chemical generic drugs), with 20 (36.4%) accelerated to market through the priority review and approval process (see Annex 2 from the original CDE news), and 2 were conditionally approved.
A total of 106 pediatric drugs were approved in 2024, including 71 marketing authorization applications. Of these, 20 (18.9%) were accelerated to market through the priority review and approval process (see Annex 3 from the original CDE news), and another 35 were approved to expand pediatric indications.
In 2024, 89 drugs were approved that were already marketed overseas but not yet in China (Class 5.1 chemical drugs, Class 3.1 therapeutic biological products, and Class 3.1 preventive biological products), with 64 being first-time approvals and 25 for adding indications to already marketed drugs (see Annex from the original CDE news).
2.2.1 The completion of the application for registration of chemical drugs
In 2024, 9,759 chemical drug registration applications were concluded. By application type, there were 1,994 clinical trial applications, 230 NDAs, 3,552 ANDAs, 1,135 bioequivalence evaluation applications, 2,449 supplemental applications, and 399 foreign-produced drug re-registration applications.
2.2.1.1 New drug clinical trial application and new drug marketing application
In 2024, 1,652 chemistry IND applications were approved, including 1,247 innovative and 405 improved-type chemical drug INDs. A total of 204 chemistry NDAs were recommended for approval, comprising 41 innovative, 58 improved-type, and 105 Class 5.1 chemical drug NDAs.
Among the approved chemistry INDs, 41.89% were for anti-tumor drugs, 11.08% for skin and ENT (Ear, Nose, and Throat) drugs, and 6.84% for endocrine system drugs. For details on the therapeutic area distribution of approved chemistry INDs in 2024, see Figure 7.
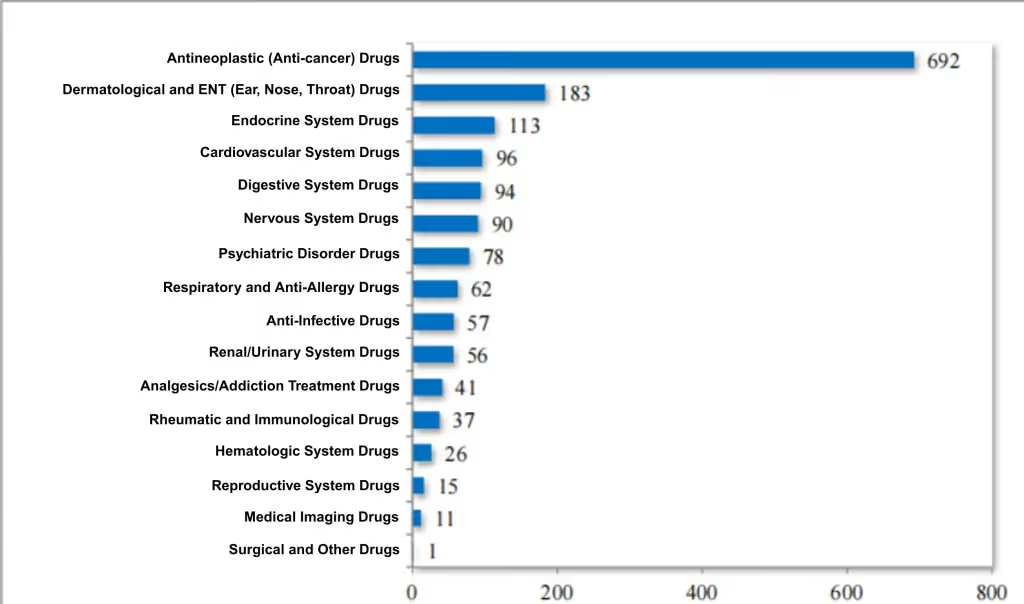
Figure 7 Distribution of indication areas for IND approval of chemical drugs in 2024 (pieces), as reported in CDE’s 2024 Drug Review Report
Among the 204 chemistry NDAs recommended for approval, 36.27% were for anti-tumor drugs, 10.78% for endocrine system drugs, and 9.31% for nervous system disease drugs. For details on the therapeutic area distribution of chemistry NDAs approved in 2024, see Figure 8.
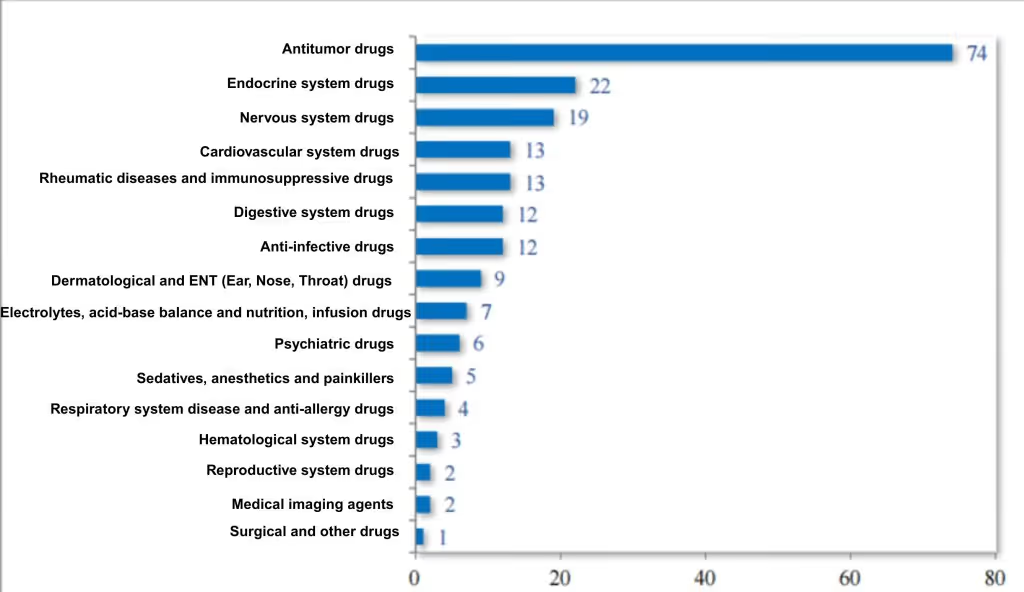
Figure 8 Distribution of indication areas recommended for approval of chemical NDAs in 2024 (pieces), as reported in CDE’s 2024 Drug Review Report
2.2.1.2 Marketing applications for chemical generics
In 2024, the CDE recommended approving 3,041 chemical generic drug marketing applications, including 289 first-to-file products, effectively meeting clinical patients' need for high-quality generics. Since the reform of the chemical drug registration classification, the CDE has cumulatively recommended approving 7,593 chemical generic drugs, covering 17 therapeutic areas such as circulation, anti-infection, and respiration. For details, see Figure 9.
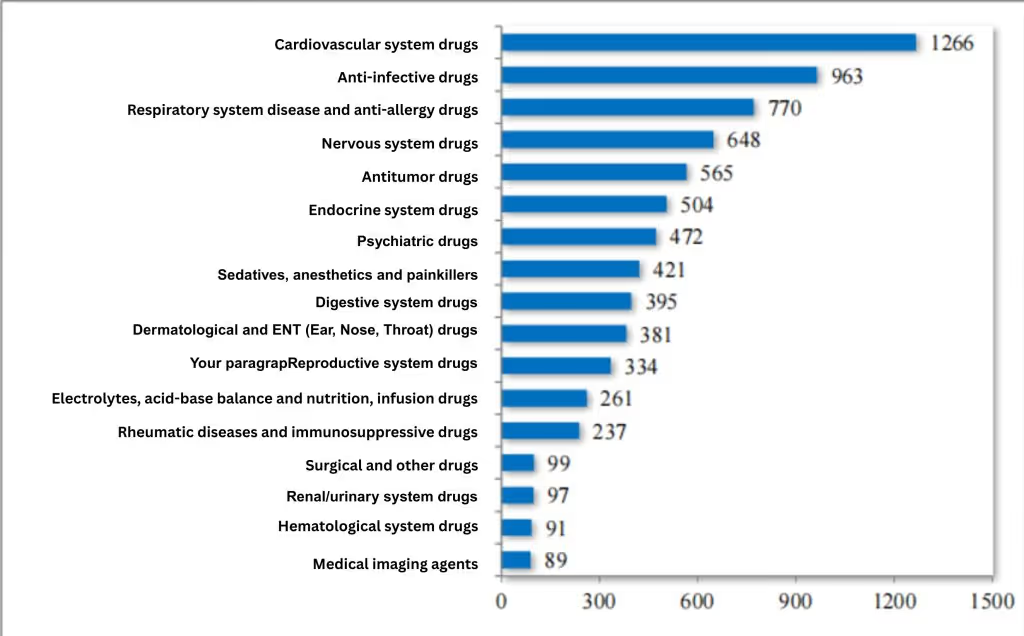
Figure 9 Distribution of ANDA indication areas for new chemical drug registration categories recommended for approval by 2024(pieces), as reported in CDE’s 2024 Drug Review Report
In 2024, the CDE recommended approving 117 drugs (21 varieties) on the national shortage list. Over the past five years, a total of 226 shortage drug applications (30 varieties) have been approved, covering 12 therapeutic areas, including circulation, electrolytes, neurology, oncology, and reproduction. For details, see Figure 10. This has effectively ensured the supply of shortage drugs.
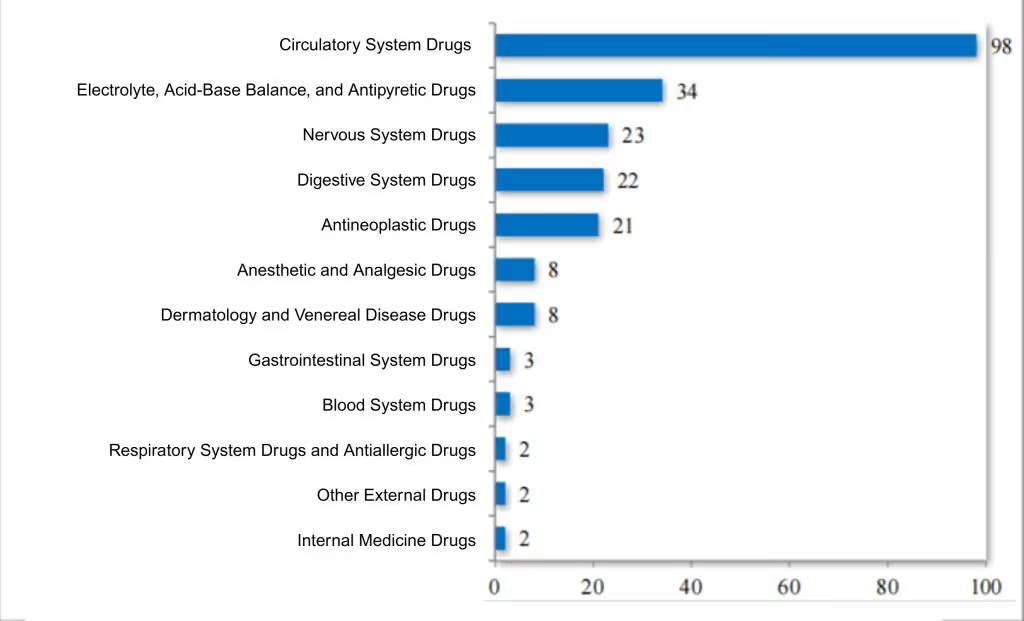
Figure 10 Distribution of drug indications in the "List of Drugs in Shortage" recommended for approval from 2020 to 2024 (pieces), as reported in CDE’s 2024 Drug Review Report
In 2024, the CDE recommended approving 17 pediatric generics (14 varieties) on the encouraged R&D pediatric drug list. Over the past five years, 59 such drugs (26 varieties) have been approved, covering 8 therapeutic areas, including neurology, oncology, and endocrinology. For specific indications, see Figure 11.
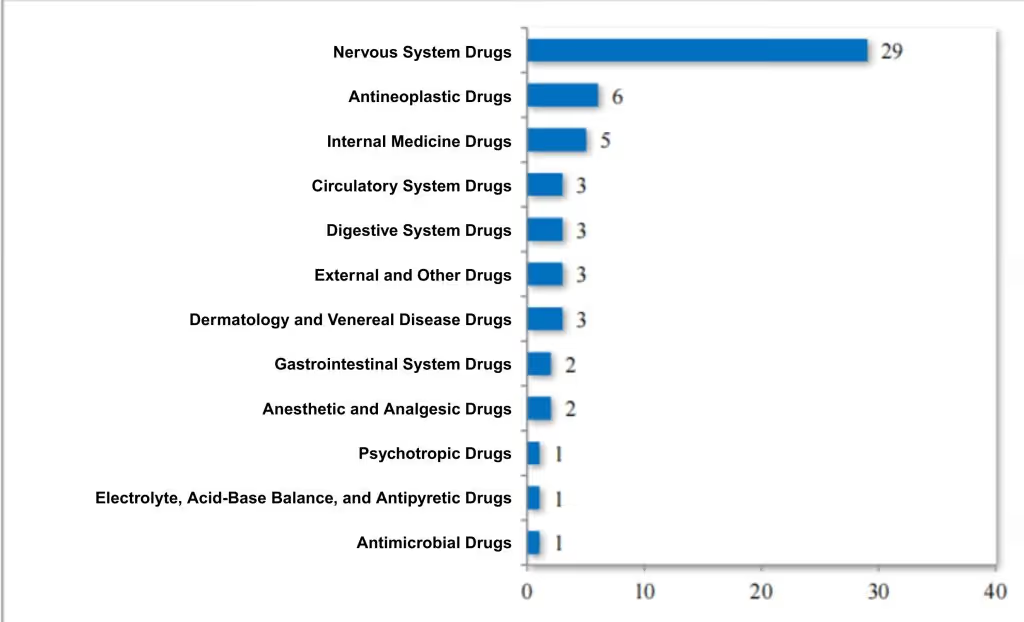
Figure 11. Therapeutic Area Distribution of Drugs Approved under the "Encouraged R&D Pediatric Drug List" from 2020 to 2024 (cases), as reported in CDE’s 2024 Drug Review Report
2.2.1.3 Consistency evaluation of the quality and efficacy of chemical generics
In 2024, the CDE continued to advance the consistency evaluation of chemical generic drugs. It released 13 batches of reference product lists, covering 929 product specifications (161 varieties). By December 31, 2024, a total of 87 batches of reference product lists had been published, involving 7,643 specifications (2,677 varieties).
In 2024, 1,011 consistency evaluation applications were approved (354 varieties), including 302 oral solid dosage forms (180 varieties) and 709 injections (174 varieties). Since the launch of the consistency evaluation initiative, a cumulative total of 4,805 applications have been approved (756 varieties), including 2,137 oral solid dosage forms (465 varieties) and 2,668 injections (291 varieties).
2.2.2 Completion of applications for registration of biological products
In 2024, 2,247 applications for registration of biological products will be completed, including 184 biological products for prevention, 2,054 biological products for therapeutic use, and 9 in vitro diagnostic reagents. According to the type of registration application, there were 1,157 IND applications, 193 NDAs, 833 supplementary applications, and 64 re-registration applications for overseas-produced drugs.
2.2.2.1 Biological products for prophylaxis
69 INDs of biological products for prophylaxis were approved for registration and classification, including 46 INDs (33 varieties) of innovative biological products for prophylaxis, 9 INDs of improved biological products for prophylaxis (7 varieties), and 14 INDs (12 varieties) of biological products for prophylactic purposes that have been marketed in China or abroad; It is recommended to approve 7 NDAs (3 varieties) of biological products for prophylactic use, which are all biological products that have been marketed for prophylactic use in China or abroad.
2.2.2.2 Therapeutic biologics
A total of 985 INDs of therapeutic biological products were approved for registration and classification, including 798 INDs (531 varieties) of innovative therapeutic biological products, 112 INDs (57 varieties) of improved therapeutic biological products, and 75 INDs (36 varieties) of therapeutic biological products marketed in China or abroad.
Among the 985 approved therapeutic biological product INDs, 55.43% were antitumor drugs, 10.66% were for dermatological and ENT (ear, nose, and throat) use, and 7.31% targeted the endocrine system. See Figure 12 for the distribution of therapeutic areas in 2024 approvals.
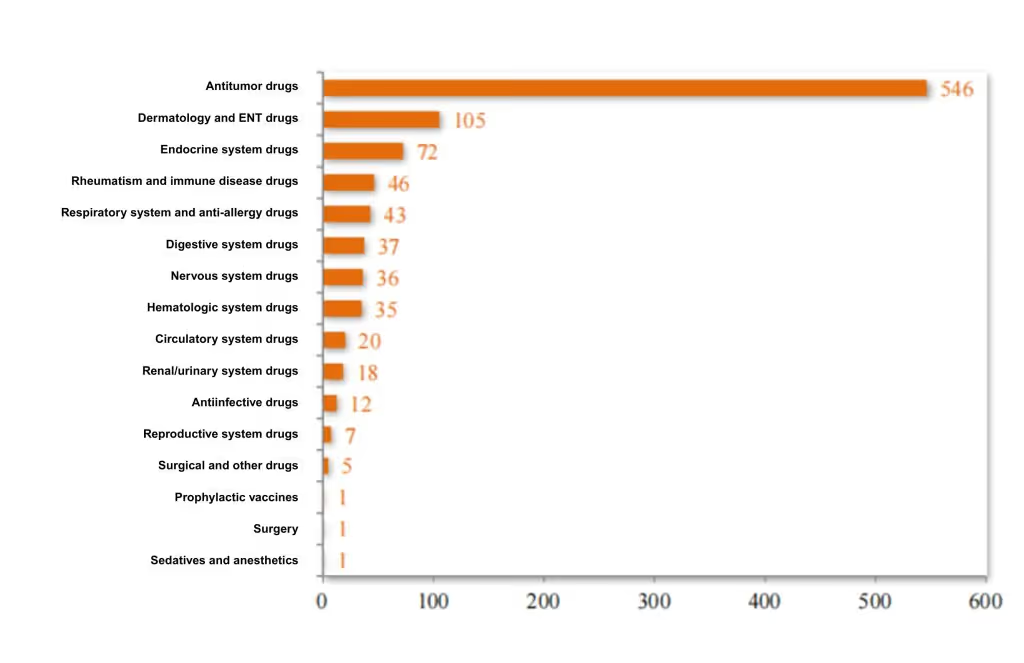
Figure 12 Distribution of INDs of Approved Therapeutic Biologics in 2024 (cases), as reported in CDE’s 2024 Drug Review Report
168 NDAs were approved for therapeutic biological products for registration and classification statistics, including 34 NDAs for innovative therapeutic biological products (24 varieties), 44 NDAs for improved therapeutic biological products (23 varieties), and 90 NDAs (49 varieties) for therapeutic biological products marketed in China or abroad, including 25 NDAs (15 varieties) for biosimilars (Class 3.3).
Among the 168 approved treatments - use biological product NDAs, 37.50% are antitumor drugs, 15.48% target the endocrine system, and 12.50% target blood-related diseases. For the 2024 distribution of approved treatment - use biological product NDAs' therapeutic areas, see Figure 13.
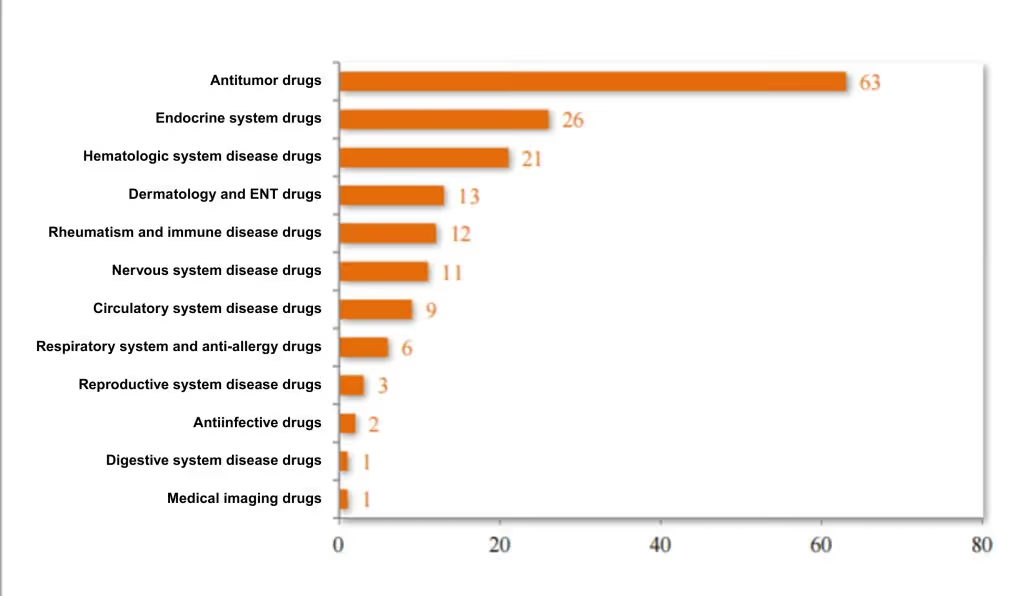
Figure 13 Distribution of Indication Areas for Recommended NDAs for Therapeutic Biologics in 2024 (cases), as reported in CDE’s 2024 Drug Review Report
2.3 The conclusion of the application for registration under direct administrative examination and approval
In 2024, 2,204 direct administrative approval-related registration applications were concluded. Among them, 1,795 were supplemental applications not requiring technical review, and 409 were temporary import registration applications.
3. Accelerated Drug Listing Registration
3.1. Breakthrough Therapy Designation During clinical trials, innovative or improved new drugs meeting specific criteria can apply for this designation. In 2024, 337 applications were received, with 91 (84 indications) approved, accounting for 27% of applications, a 30% increase from 2023. Since its implementation in 2020, 294 applications (246 indications) have been approved, mainly in oncology, neurological, and anti-infective drugs. These drugs receive priority communication and support during trials to speed up development.
3.2. Conditional Approval Drugs for life-threatening diseases without existing treatments, or those urgently needed for public health, can be conditionally approved if certain requirements are met. In 2024, 37 applications (27 indications) were conditionally approved, including 18 new indications and 9 added to already marketed drugs. Meanwhile, 9 conditional approvals turned into full approvals after confirmatory trials. Since 2020, 187 applications (135 indications) have been conditionally approved, with oncology drugs being the largest group.
3.3. Drugs with significant clinical benefits, such as urgently-needed shortage drugs, and innovative drugs for major infectious and rare diseases, can apply for priority review and approval. In 2024, 124 priority review and approval applications (88 drug varieties) were accepted, up 14.81% year-on-year. Of these, 110 applications (74 drug varieties) were approved, up 29.41% year-on-year. Since 2020, 496 applications have entered this procedure, with anticancer drugs being the most numerous.
4. Drug Development and Communication
4.1. Communication Meeting Applications and Processing In 2024, the Drug Evaluation Center received 5,554 communication meeting applications and processed 4,912, serving 1,575 enterprises and 3,551 drugs. There were 691 face-to-face or phone meetings, up 12.91% year-on-year. Among processed applications, 56.05% were Category Ⅱ meetings at key development stages, such as pre-IND and pre-NDA meetings, helping businesses get professional guidance and optimize development plans.
4.2. General Technical Consultations In 2024, 16,156 general technical questions were received, with 15,449 answered. The center published seven batches of 31 common question answers on its website, covering registration acceptance and technical review, totaling 240 common questions. These measures enhanced review transparency and consistency, helping businesses better understand requirements and reduce development uncertainties.
Further Information
To read the original announcement from CDE 2024 Drug Review Report with the detailed drug registration and regulatory trends in China, please click here.
Need support for China marketing authorisation applications or other advice? Please reach out to us at Cisema to speak.
GET IN TOUCH
🌐 Send us your enquiry📚 Request our whitepapers📣 Sign up for our newsletter



