News

The NMPA (National Medical Products Administration) announced the 2020 project plan and selection process for 86 medical device industry standards.
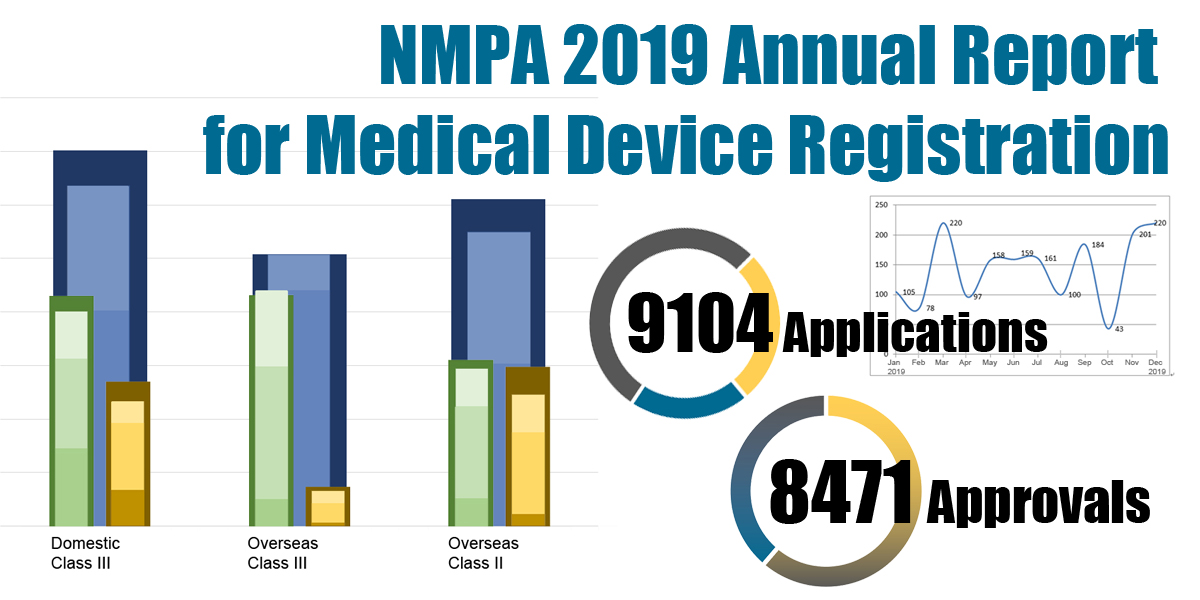
On 18.03.2020, the NMPA (National Medical Products Administration) issued the 2019 Annual Report for Medical Device Registration.In 2019, the NMPA received a total of 9,104 applications for the initial registration, registration renewals and changes in licensing items of Class III (Domestic and Overseas) and Class II (Overseas) medical devices, an increase of 37.8% from 2018. Amongst the 9,104 applications, the NMPA approved a total of 8,471 applications with an increase of 53.2% as compared to 2018. Also, the NMPA handled a total of 1,383 filing applications of imported Class I medical devices, a decrease of 20.7% as compared to 2018.
Table 1. NMPA - No. of Applications and Approvals for Medical Devices and IVDs
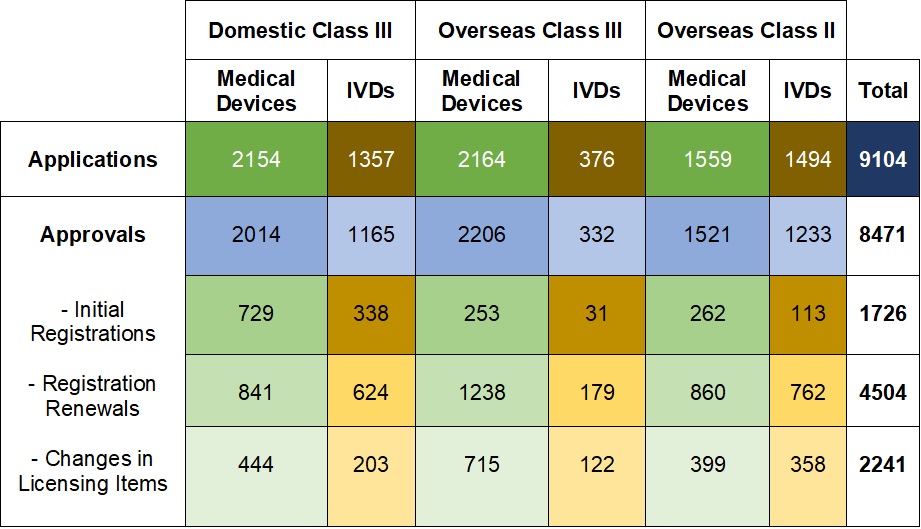
Chart 1. NMPA – No. of Applications and Approvals for Medical Devices and IVDs
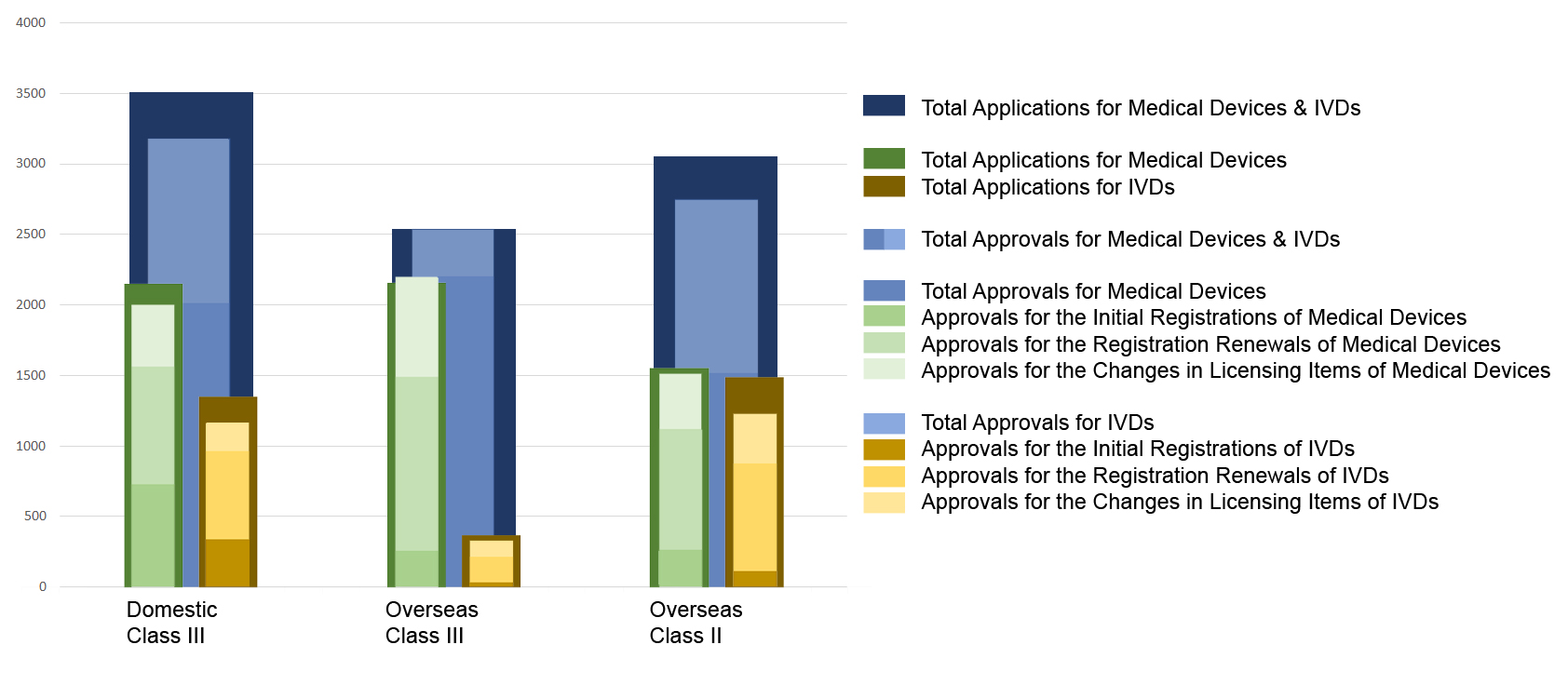
Chart 2. Percentage Distribution of Three Types of NMPA Approvals
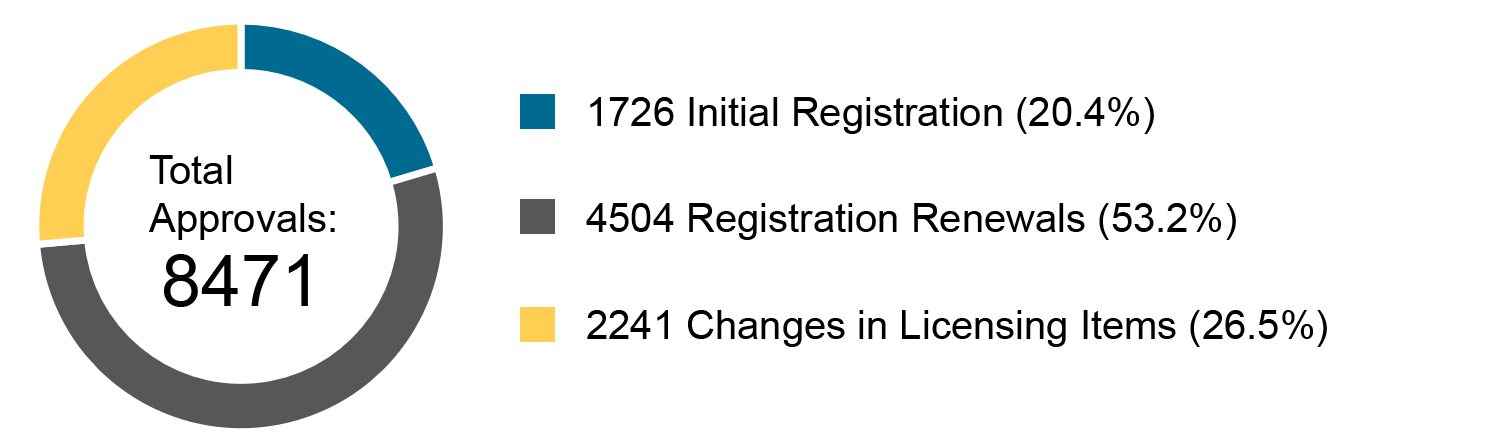
Chart 3. Percentage Distribution of Approvals for Medical Devices and IVDs

Chart 4. NMPA – No. of New Registrations Approved from January 2013 to December 2019
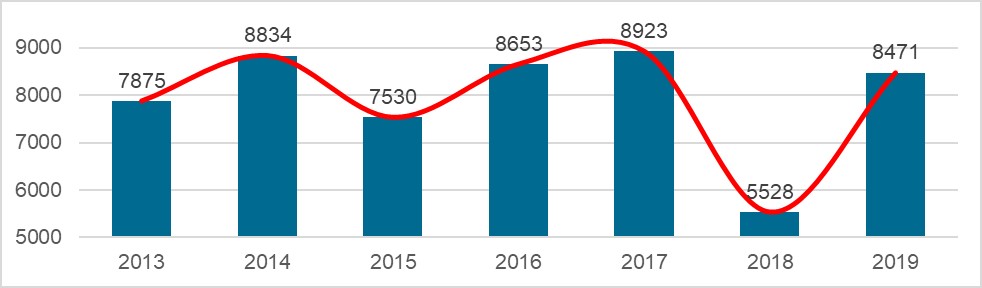
Chart 5. NMPA – No. of New Registrations Approved from January 2019 to December 2019

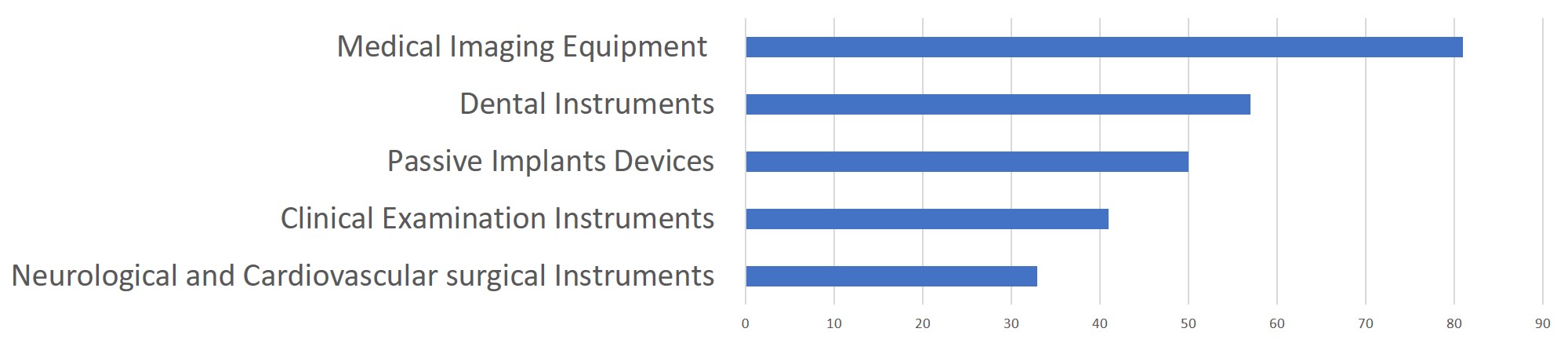
In 2019, the NMPA approved 1,726 new registrations in total.The provincial medical product administration (MPAs) authorities approved a total of 17,017 applications of China domestic Class II medical devices, an increase of 53.4% from 2018.The municipal medical product administration (MPAs) authorities handled a total of 16,754 filing applications of China domestic Class I medical devices, decreased by 2.4% as compared to 2018.China continues to focus on the supply of high-end, high-cost medical devices from abroad. The top five class II and III product groups of foreign origin to be registered in 2019 were:1. Medical Imaging Equipment (81 Registrations)2. Dental Instruments (57 Registrations)3. Passive Implants Devices (50 Registrations)4. Clinical Examination Instruments (41 Registrations)5. Neurological and Cardiovascular Surgical Instruments (33 Registrations)
Chart 6. 2019 Top 5 Product Groups - Registration of Overseas Class II and III Medical Devices
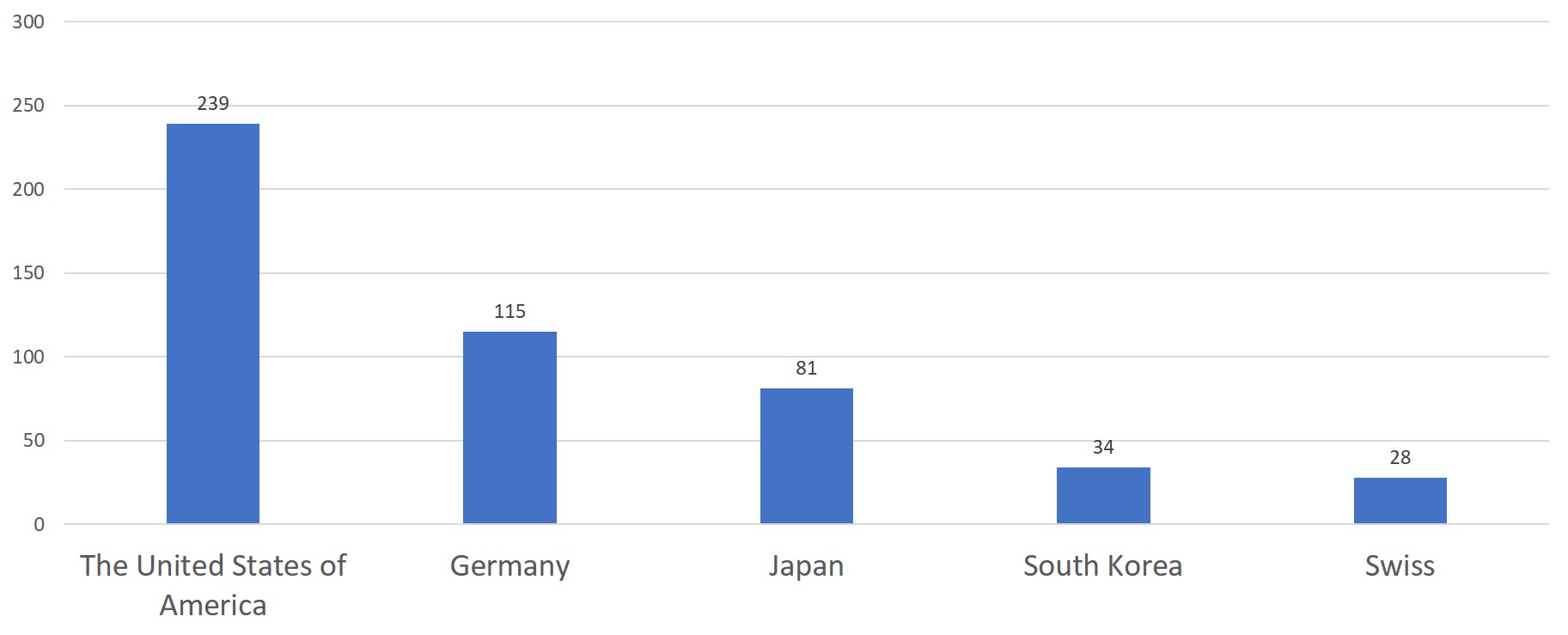
The United States of America, Germany, Japan, Korea and Switzerland have the highest number of initial registrations for overseas medical devices, taking up to 75.4% of the total number of initial registrations for overseas medical devices.
Chart 7. 2019 Top 5 Countries - Exporting Medical Devices to China
Special Review Procedures for Innovative Medical DevicesNMPA received a total of 179 applications for special review and approval of innovative medical devices, including 31 applications for priority review. A total of 19 innovative medical devices were approved:Imported1) Micra Transcatheter Leadless Pacemaker systemDomestic2) PET/CT imaging system3) Nucleic acid amplification detection analyzer4) Decellularized corneal implant5) Left atrial appendage occluder system6) Abdominal aorta stent-graft and delivery system7) Bioabsorbable coronary rapamycin-eluting stent system8) Porous tantalum bone filling material9) Patient monitor10) Left atrial appendage closure system11) Intensity-modulated radiotherapy planning system software12) Digital Mammography System13) Transcatheter aortic valve system14) Single-use intravascular imaging catheter15) Non-Invasive Blood Glucose Meter16) Implantable left ventricular assist system17) Coronary angiographic blood flow reserve fraction measurement system18) Disposable invasive pressure sensor19) Positron emission and X-ray computed tomography scanning systemFurther information concerning this topic can be obtained from:Cisema (Hong Kong) LimitedTel.: +852 3462 2483info@andrewb655.sg-host.comwww.cisema.com/en

According to the Guide, the registrant of the medical device is responsible for implementing a suitable QMS for the product realization process.

The NMPA announced new administrative measures designed to increase the post-market surveillance applicable to all registered medical devices sold in China.

The principles describe the manufacturing requirements to ensure the medical device operates safely and performs as intended。

AUSSENWIRTSCHAFT AUSTRIA and Cisema cohost the webinar to talk about the regulatory changes for both medical device and cosmetic industries.

Medical devices approved by the NMPA or the Ministry of Food and Drug Safety of Korea can apply for the MDACS listing in Hong Kong within a trial period

The prevention and control work were deployed as the top priority, and the registration process of some overseas medical products was expedited.

NMPA provides a regulatory pathway for CMDs to meet the special clinical demands for medical institutions in China to treat designated patients.

Cisema will submit a proposal to the NMPA about the draft regulation before the consultation period ends on 31.03.2020. If you are interested to contribute, please provide your input to us

On 4.3.2020, the China-Britain Business Council (CBBC) and Cisema held a webinar together to talk about the market, regulations and standards for overseas medical device products to enter the China market. Our business consultant, Anna King, provided an overview of the approval systems for NMPA registration in China and the recent implementation of urgent measures to expedite the approval procedures for medical and health products due to the CoronaVirus outbreak.Watch the recording of the webinar on Youtube.

New product certification regulation from organization CCAP for automobile lighting, external plastic parts and break disks

The Israel Export Institute co-hosted a webinar together with Cisema to introduce the China administration bodies, regulations and standards for cosmetics.

Currently, there are 14 municipalities with the capacity to file non-special purpose cosmetics, and Cisema can guide you through the MPA filing process.

On 04.02.2020, the authorities SAMR (State Administration for Market Regulation) and CNCA (China National Certification Administration) published in their Notice No 9/2020 general support measures to contain the corona virus outbreak. Chinese certification organizations are bound by these requirements.The measures include:
- Processes are to be optimized so that personal contact is reduced as much as possible.
- Factory inspections by a Chinese certification organization, which must be postponed due to the current situation, must be communicated openly with the affected certificate holders in order to fix an alternative date by mutual agreement. Or other solutions are agreed upon.
- Applications for factories that have stopped production due to the outbreak and are unable to complete an inspection of their manufacturing plant in order to obtain the desired certification (e.g. CCC - China Compulsory Certification), but are subject to a time limit for this, may not be suspended. The necessary inspection should be completed at the latest 3 months after the official announcement that the state of emergency has been lifted.

The Ministry of Commerce announced the list of intermediary services and administrative examinations and approvals for import and export of goods in China.

Cisema has the capability to help you comply with Hong Kong MDACS requirements and expand your businesses in the Greater Bay Area

On 27.12.2019, the China Ministry of Finance, General Administration for Market Regulation and 11 other ministries announced (No.96-2019) the List of Cross-border E-commerce Retail Imported Commodities (2019 edition) covering a total of 1413 types of commodities. The list added roughly 100 types of goods, and removed 8 types from the 2018 edition. According to China Customs, products on the list are exempted from submitting related import licenses. When entering the special trade zone, the commodites on the list will be checked by customs in accordance with the standard procedures for clearing commodities, but will then be exempted when leaving the special trade zone. The updated list came into effect on 01.01.2020.On 24.12.2019, the Chinese State Council announced (No.36-2019) that the State Taxation Administration had approved the establishment of 24 pilot cross-border e-commerce (CBEC) zones - in addition to the 35 pilot CBEC zones that have previously been established - and introduced exemptions on value-added tax and consumption tax for retail and exported goods. The approved document announces the initial plan of how the trial zones will be constructed and replicated across China. More detailed information regarding tax rebates, government support, and international logistic services are expected to follow after this announcement.Please do contact us to know if your products are involved.

On 06.11.2019, the China NDRC (National Development and Reform Commission) announced (No.20-2019) the prohibition of daily chemical articles containing plastic microbeads for production after December 31, 2020, and for sale after December 31, 2022.Microbeads are small plastic particles, mostly included in cosmetic and cleaning products, and are known to cause water pollution to the marine and freshwater environments. Currently, 14 states have already taken action towards the ban on microbeads. Although the details of the ban for production and sales in China is still subject to further refinement, brands and manufacturers are expected to reformulate their products containing microbeads, while some have already replaced microbeads with natural abrasives as an alternative in daily chemical articles.The announcement (No.20-2019) is based on the Industrial Structure Adjustment Guidance Catalogue (2019 Edition) that was approved at the 2nd committee meeting on 27.08.2019, and has replaced the 2011 version since 01.01.2020.

On 20.12.2019, the China NMPA (National Medical Products Administration) announced (No.91-2019) that it would add 148 medical devices and 23 in-vitro diagnostic reagents to the clinical trial exemption list, as well as to revise the names and descriptions of 48 medical devices and 4 in-vitro diagnostic reagents. Medical devices and in-vitro diagnostic reagents on the clinical trial exemption list won’t require completion of clinical trials in order to achieve NMPA certification.This new clinical trial exemption list is effective since the date of announcement on 20th December.Feel free to contact us if you would like to know what products were included in the clinical trial exemption list for medical devices, and the clinical trial exemption list for IVDs.
Get in Touch with Our Consultants
And discover how we can support you.
