Blog Posts

Understand MFDS clinical trial requirements for digital medical devices, including real-world evidence, data-based studies, and software validation expectations.

Supporting foreign manufacturers entering South Korea with regulatory services for medical devices, IVDs, and quasi-drugs.

Understand Indonesia halal certification requirements for health supplements, including BPJPH registration, ingredient review, and halal labeling obligations.

Learn which products are covered by Indonesia's halal framework, the labeling and manufacturing obligations, and phased compliance timelines.

Learn how Indonesia halal cosmetic certification works, including BPJPH requirements, halal labeling rules, ingredient compliance, and registration obligations.

Cisema offers regulatory insight and market access strategy for medical devices in Thailand, Indonesia, and the Philippines.

Learn how to register medical devices in Indonesia, including Authorized Representative rules, classification, dossiers, timelines, and fees.
%20Market%20A%20Guide%20for%20Foreign%20Manufacturers%20(1).avif)
Understand the pathway for intraocular lens (IOL) registration in China, including NMPA requirements and key compliance steps.

China’s 15th Five-Year Plan signals stronger support for innovative drugs, advanced medical devices, reimbursement reform, and NMPA regulatory pathways.
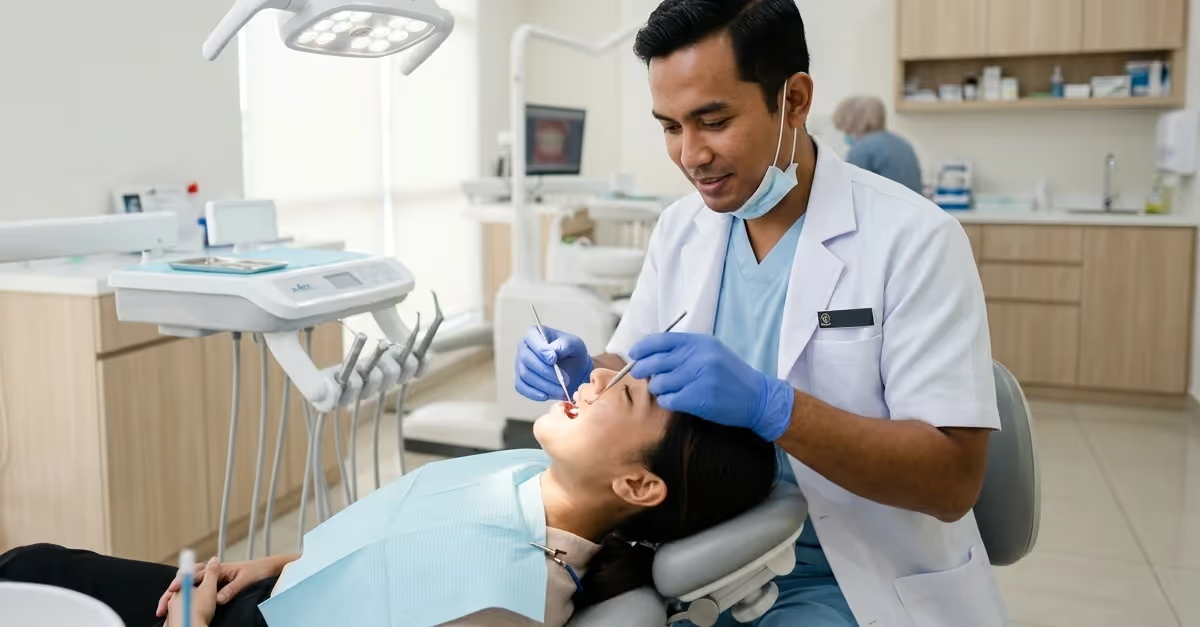
Enter Southeast Asia’s dental market with a clear strategy: navigate regulatory complexity, sequence market entry, and retain control of registrations to scale efficiently.
.avif)
Learn about Cisema’s redesigned website with faster performance, clearer navigation and expanded Asia Pacific coverage.

A complete guide to China hyaluronic acid registration, covering classification, NMPA requirements, and compliance steps for manufacturers.
Track all imported Class II and III medical device approvals in China with this monthly updated NMPA submission tracker.

Learn how Malaysia classifies drug–medical device combination products and how the PMOA determines pathways and compliance responsibilities.

Practical China medical device glossary explaining NMPA terms, with clear comparisons to US FDA and EU MDR terminology.

This guide explains Singapore’s HSA regulatory system and why it is a strategic first step for entering Southeast Asian markets with medical devices, pharmaceuticals, and cosmetics.

Learn how a smart clinical trial strategy for medical devices in China can speed up NMPA approval and bring your product to market faster.
.avif)
Learn 5 key steps for China dental medical device registration, covering classification, biocompatibility testing, and legal agent selection.

Get answers to common China medical device and IVD registration questions, from NMPA classification rules to testing and renewal requirements.

Gain clarity on often-misunderstood China Manufacture License (CML) requirements, including SELO approval, demo products, and CSEI audits.
Get in Touch with Our Consultants
And discover how we can support you.
