News

CNCA issued 11 trial CCC certification implementation rules for fuses, electric tools, welding machines, automotive components, and safety glass.

China’s MIIT and nine ministries introduce a new AI ethics review and services framework establishing tiered oversight, compliance obligations, and governance pathways for AI activities in China.

China issues new guidelines on medical device adverse event risk evaluation and periodic report review, tightening post-market surveillance requirements
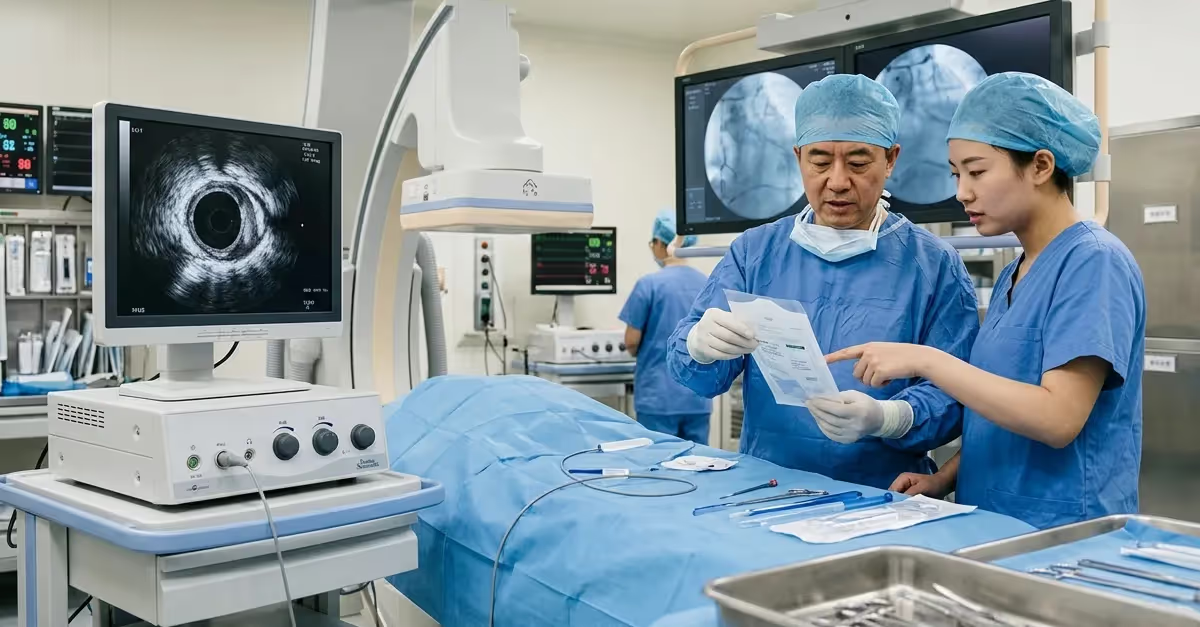
China’s CMDE releases a new IVUS registration review guideline, defining technical, clinical, and testing requirements for NMPA medical device registration

Malaysia’s MDA publishes the 7th edition of guidance on medical device labelling, with new e-labelling, e-IFU, and accessibility requirements

Vietnam’s Drug Administration (DAV) is stepping up cosmetic product recalls as post-market surveillance tightens.

BPOM confirms pharmaceutical authorization, GMP oversight, and regulatory authority remain unchanged under the Indonesia–US trade agreement.
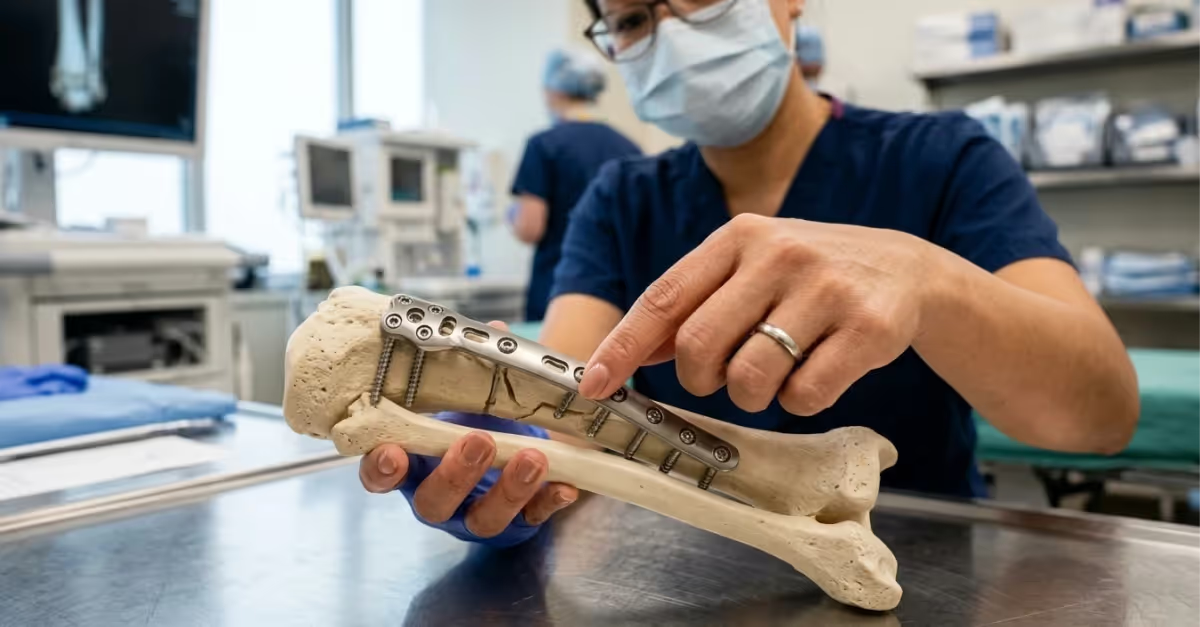
The 26 new medical device standards cover orthopedic implants, IVDs, active devices, and software risk management.

China’s NMPA has opened consultation on a draft mandatory toothpaste safety standard that could tighten product requirements.

China's HAF003 reform and participation in the "Tripling Nuclear Energy Declaration" signals stricter compliance and new opportunities.

China’s NMPA mandates full UDI coverage for all medical devices by 2029, linking compliance to registration, reimbursement, and market access.

China’s NMPA issues new guidance on clinical trial protocol pre-review for innovative devices, impacting overseas fast-track approvals.

China clarifies GACC Order 280 rules for imported food registration and customs declarations ahead of June 2026 enforcement.

China NMPA adds four new cosmetic testing methods effective July 2026, impacting compliance and safety assessment strategies.

China’s NMPA updates cosmetics testing methods, adding CBD detection and tighter impurity controls impacting global compliance.

China releases guiding principles for benefit-risk assessment using multi-regional clinical trial data to support global drug development.

China releases new subject selection guidance for anti-tumor drug bioequivalence and pharmacokinetic studies.

China’s CCC mark pilot reform requires traceability QR codes for power banks, electric bicycles, and gas appliances.

SAMR introduce stricter recall and compliance requirements for enterprises involved in cross-border e-commerce retail imported food.

China NMPA’s 2025 report highlights growth in medical device approvals, including advanced imported devices.
Get in Touch with Our Consultants
And discover how we can support you.
